Look for signs of ‘Black Death’
Penaeid shrimp including important commercially cultured species such as tiger shrimp (Penaeus monodon), white shrimp (Litopenaeus vannamei), blue shrimp (L. stylirostris) and kuruma prawn (Marsupenaeus japonicus) require dietary vitamin C for maximum growth and survival. Guary et al., 1976 were the first to observe that vitamin C was required for maximum growth in penaeid shrimp. In 1977 and 1979, a vitamin C deficiency disease of penaeid shrimp was described (Lightner et al., 1977: Magarelli et al., 1979).
Deficiency symptoms
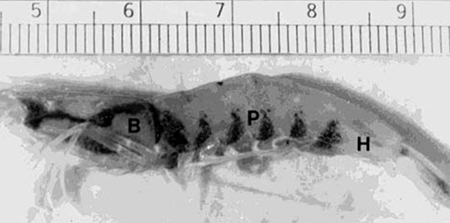
Vitamin C deficiency disease (often called Black Death), is characterized by black melanized lesions of the loose connective tissues of the body, especially under the carapace, the abdomen, the gills, and in the foregut and hindgut. Gross lesions may show a series of black bands at the articulations of the abdominal segments, giving the affected animal a striped appearance (Fig. 1).
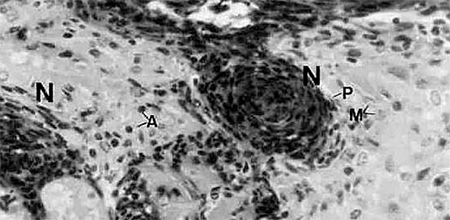
Histological examination reveals multiple melanized lesions composed of hemocytes and associated with inflammatory cells (Fig. 2). In experimentally induced vitamin C deficiency, shrimp with disease signs and mortalities occur starting in weeks 3-5. Daily mortalities in a deficient population may reach 1 to 3 percent per day, and some but not all mortalities will display Black Death signs. Once an animal shows Black Death signs, cessation of feeding and death invariably occur within 24 to 72 hours. Without addition of dietary vitamin C, mortalities may approach 100%. However, if vitamin C is added to the diet of the deficient population, the disease and resulting mortalities may be arrested.
Physiology
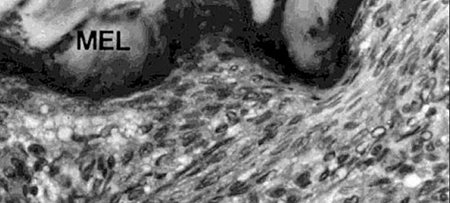
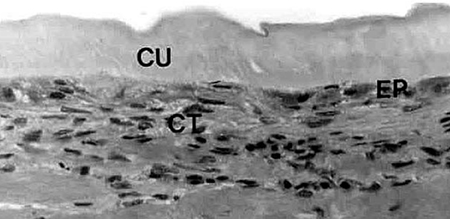
In 1979, Hunter et al. were able to show that shrimp, like other vertebrates, require vitamin C for proline hydroxylation of collagen. In growing shrimp without access to vitamin C, the collagen support structure of the shrimp is not properly formed, which leads to the disease signs previously described. Hunter (1979), and Lightner et al. (1979), showed that dietary vitamin C was also required for wound repair of penaeid shrimp, another role of structurally sound collagen formed by shrimp with adequate vitamin C supply (Figures 3 and 4).
Quantitative requirement
Early researchers studying shrimp vitamin C concluded that the requirement was as high as 3,000-10,000 PPM. These studies were conducted using crystalline vitamin C (ascorbic acid), and much of this form was destroyed by pelleting or leached from the diet prior to consumption. Estimates of minimum vitamin C requirements for penaeid shrimp determined using stable phosphorylated forms are much lower, due to the stability of ascorbyl phosphates to oxidation and to their limited water solubility. Estimates for ascorbyl phosphate requirements in penaeid shrimp have ranged from 40 to 430 ppm (Shigueno and Itoh, 1888; He and Lawrence, 1993; Shiau and Hsu, 1994).
Benefits of elevated doses
Other nutritional research has shown that vitamin C serves a protective role against disease and physiological stress at dietary inclusion levels which are often higher than the levels established for growth requirement. Table 1 summarizes the effect of dietary vitamin C on survival of penaeid shrimp subjected to physiological and pathogenic challenges in lab trials.
Takahashi et al. (1989) found that 200 ppm AP resulted in increased survival of M. japonicus following an injection of pathogenic Vibrio compared to a no-vitamin-C control. They also reported impaired phagocytosis in vitamin- C-deficient M. japonicus. Kanazawa (1995) also found an improvement in survival of M. japonicus fed AP following injected Vibrio challenge compared to non-supplemented shrimp. Merchie et al. (1995) found that dietary AP enhanced survival of L. vannamei during a natural epizootic of Vibrio harveyi. Supplementation of shrimp diets with 20 ppm to 100 ppm APP enhanced survival compared to a no-vitamin- C control. Survival was much higher for shrimp receiving 2,000 ppm of AP.
At least four studies have shown that dietary vitamin C affects survival of shrimp following osmolarity stress, a common occurance in shrimp ponds after heavy rain. Merchie et al. (1995a), found L. vannamei post-larvae fed 0 or 20 ppm of AP had lower survival than those fed 40 or 100 ppm of AP following instantaneous salinity drop to 0 ppt. The highest survival was achieved when shrimp were fed 2,000 ppm of AP. In a similar osmolarity study, Merchie et al. (1995b) found P. monodon post-larvae survival higher for treatments receiving 200 ppm AP or 2000 ppm AP than for those receiving 0, 20 or 40 ppm AP.
A second confirmatory trial documented the effect of vitamin C on survival of P. monodon post-larvae subjected to 0 ppt salinity (Merchie et al., 1996). Survival was highest for shrimp receiving 3,400 or 1,700 ppm AP, compared to shrimp receiving 100 ppm AP. A relation between dietary vitamin C intake and survival of juvenile P. monodon following osmolarity stress was also found (Hunter et al., 1998). Survival was greatest for shrimp receiving 189 ppm AP, intermediate for shrimp receiving 53 ppm AP, and lowest for shrimp receiving no vitamin C following gradual salinity reduction to 0 ppt.
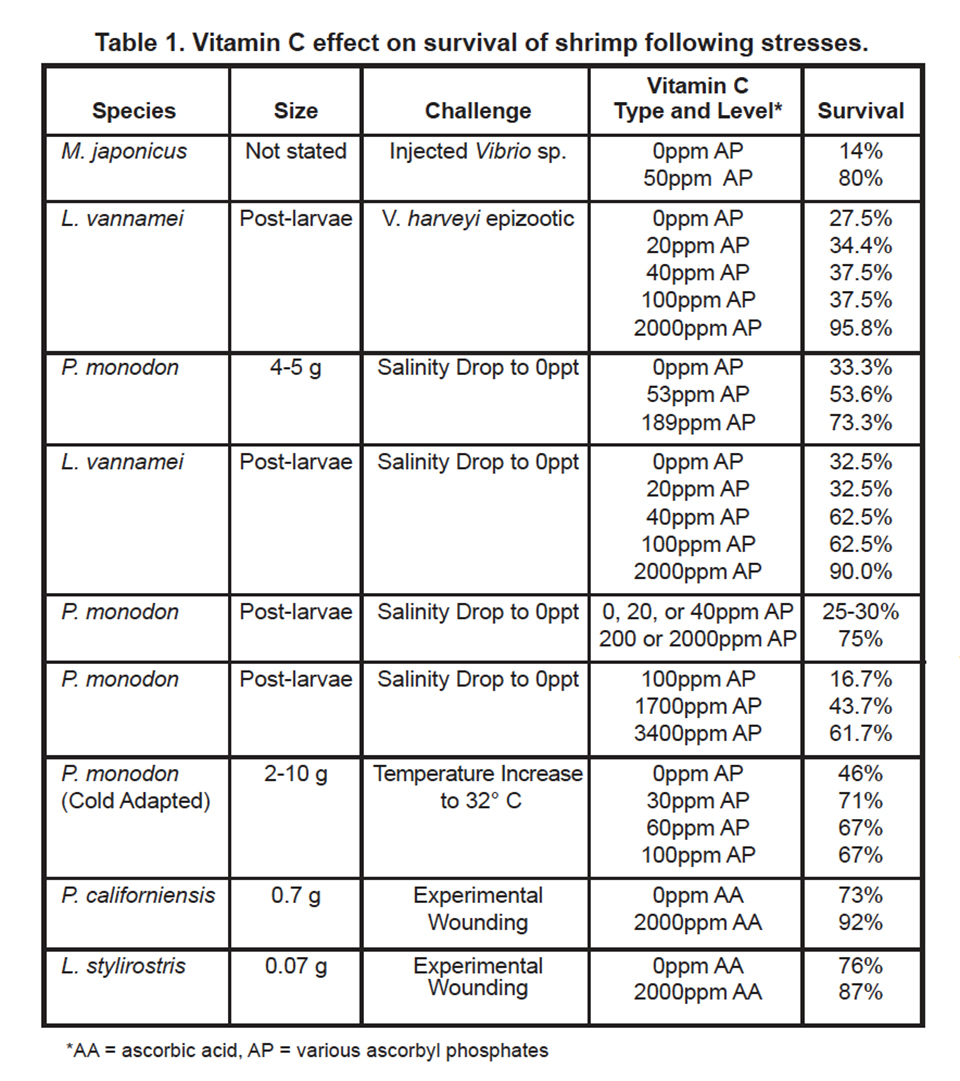
Survival of penaeid shrimp following temperature stress or experimental wound healing was also improved following dietary vitamin C supplementation. Machigashira et al. (1991) found that cold-adapted P. monodon survival following temperature increase to 32 degrees-C was higher for treatments receiving 30, 60 or 90 ppm AP compared to the no vitamin C treatment. Hunter (1979) and Lightner et al. (1979) found that survival of P. californiensis juveniles and L. stylirostris juveniles were higher following experimental wounding for shrimp fed a diet with 2,000 ppm added AA compared to shrimp fed a diet without added AA.
Conclusions
Vitamin C is required by shrimp for proper collagen formation and wound repair. Severe deficiency can result in “Black Death” syndrome, which is characterized by black melanized lesions. The vitamin C requirement for shrimp was originally overestimated due to use of crystalline forms of vitamin C, which are susceptible to high losses due to feed processing, storage, and leaching. The estimated requirement using stable phosphorylated forms ranges from 40 to 430 ppm for various species and life stages. Elevated dietary levels of vitamin C have been shown to improve resistance to disease, salinity shock, and other stresses.
(Cited references may be requested from the author.)
(Editor’s Note: This article was originally published in the April 2000 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Brian Hunter
Manager, Roche Aquaculture Centre Asia Pacific
Rovithai, Ltd.
11/F, 2535 Sukhumvit Rd.
Bangkok 10250 Thailand[109,111,99,46,104,116,46,99,115,107,64,114,101,116,110,117,104,98]
Tagged With
Related Posts

Health & Welfare
Colombian shrimp research associates TSV, NHP selection, resistance
In an NHP (necrotizing hepatopancreatitis) challenge test, shrimp from a Colombian breeding program had higher resistance and 30 percent greater survival than a control line of Taura syndrome virus-free shrimp.
Health & Welfare
Study: Oral method vaccinates rainbow trout against IHNV
Working on a system for oral delivery of IHNV vaccine via fish feed, researchers' goal is to adapt a proven oral DNA-based vaccine given to salmon in Canada to rainbow trout through encapsulation with a polymer or liposome.
Aquafeeds
Artificial microparticles for delivery of nutrients to marine suspension-feeders
Commercial development of artificial microparticles should reduce reliance on live feeds and lead to less expensive diets.
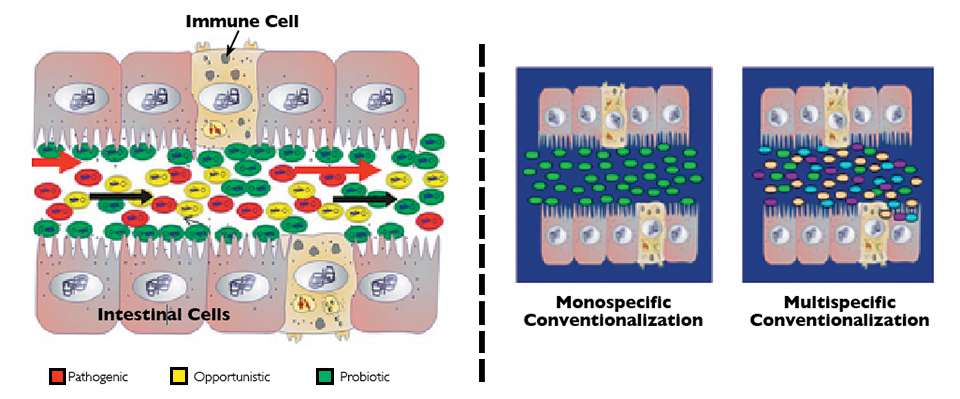
Health & Welfare
Domestication of gut microbiota can improve shrimp aquaculture
In studies, bacteria and yeasts isolated from the native microbiota of wild shrimp at larval, juvenile and adult life stages were administered to early-stage shrimp via the culture water.


