Proven record of success in hatcheries

Bacterial probiotics are living organisms that are utilized to improve disease resistance, water quality, and/or nutrition. Depending on the mode of action, probiotics can be added to the culture water or the diet.
For example, by adding the probiotic CA2 to culture water, oyster larvae – through efficient filter-feeding – get the bacterial nutrients needed. But in order to use digestive probiotics with adult fish, it is better to mix the probiotic with food to maximize the probiotic contact in the animals’ digestive systems.
Probiotics have a proven record of success in hatcheries where they are added to clean water with no preexisting microbial community. However, results have been unreliable in ponds where they are added to natural waters with established microbial communities.
The keys to success of probiotic addition (“bioaugmentation”) are selection of the right “bugs” and provision of appropriate environmental conditions (“microbial management”). The following case studies illustrate probiotic procedures used succesfully in hatcheries.
Case studies
Larval cultures of Pacific oyster (Crassostrea gigas)
An Alteromonas strain (CA2) was found to increase the growth of oyster larvae by 39 to 56 percent through supply of nutrients deficient in algae. The use of this probiotic under hatchery conditions lead to a consistent improvement in larval production.
The microbial management consisted of disinfecting tanks and screens, 1-μm filtration of seawater to remove protozoa, the addition of probiotic at each water change at 105 to 106 cells per milligram, and holding the culture water temperature above 25 degrees-C.
Larval cultures of marine fish red drum (Sciaenops ocellatus)
An elaborate microbial management was developed, where all sources of microbes were eliminated and the microbial niches were seeded with bacterial probiotics. Using this management, a blend of four probiotic strains enhanced the growth rates of rotifers 1.7 to 4.7 times, while improving survival of fish from 23.7 percent to 67.3 percent.
Microbial management involved disinfecting culture systems with chlorine, disinfecting compressed air with filter cartridges, disinfecting all culture seawater by filtration and chlorination, disinfecting red drum eggs with hydrogen peroxide, disinfecting rotifer cysts with chlorine, culturing bacteria-free algae with bacterial probiotics, culturing bacteria-free rotifers with microbial probiotics, and culturing fish larvae with the probiotic-enriched rotifers.
Implementation of these management steps improved larval survival, and also produced two other benefits:
- No culture crashes occurred in systems treated with bacterial probiotics.
- The notorious differences in production observed between replicate tanks in cultures of algae, rotifers, and fish larvae (quantified as coefficients of variation) were significantly reduced by microbiological management.
Larval cultures of crab (Portunus trituberculatus) in Japan
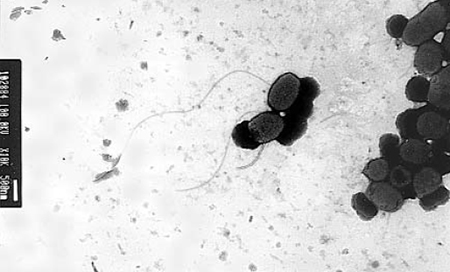
The strain Thalassobacter utilis (PM-4) (Fig. 1) improved survival of crab larvae while repressing the growth of the pathogenic bacteria Vibrio anguillarum and the fungus Haliphthoros sp. In 33 trials reported, use of the bacterial probiotic increased the survival rate of crab larvae from 15.6 percent to 28.3 percent, and the number of trials that completely failed was reduced from 12 in the controls to one under the bacterial treatment. The management included 400 μm-filtration and chlorination of seawater, and bacterial probiotic added daily for eight days at 105 to 106 cells per milligram.
Dr. Masachika Maeda, who developed the procedure, cautions against the danger of omitting a single application of the probiotic, as it could result in total loss of the larval culture due to population explosions of Vibrio. He also found a Pseudoalteromonas undina strain (VKM-124) (Fig. 2.) that repressed the activity of the Sima-Aji Neuro Necrosis Virus (SJNNV) that infects jacks. With microbial management similar to that used for PM-4, Maeda obtained efficient biocontrol of the viral agent using the VKM-124 strain.
Larval cultures of shrimp (Litopenaeus vannamei) in Ecuador
David Garriques reported in 1995 that a strain of Vibrio alginolyticus improved shrimp survival from 74.5 to 90.1 percent and decreased the concentration of a pathogenic strain of Vibrio parahaemolyticus. He recently confirmed that the total yield of the four hatcheries at El Rosario Granjas Marinas (200 million PL per year) is produced with the bacterial probiotic. Currently, the market price from diverse laboratories in Ecuador is U.S. $1.60 to $2.00 for 1,000 PLs, while El Rosario charges between U.S. $2.40 and $3.50 for its probiotic-grown larvae.
The microbial management applied consisted of 5-μm filtration, activated charcoal filtration and chlorination of culture seawater, bacterial probiotic prepared fresh every day and added at 105 cells per milligram daily from stage nauplii 5 to PL1, and algal cultures started from axenic clones and grown with aseptic techniques.
Conclusion
Consistent beneficial results with bacterial probiotics have been obtained in hatcheries that have utilized appropriate strains coupled with effective management. Microbes should be selected based on previously determined effectiveness with the target organisms and culture conditions. Quality control of the probiotic is important to eliminate contamination and assure viability, counts and activity. Effective management involves provision of favorable conditions for probiotic activity, such as removing bacterial predators, reducing or removing microbial competition, avoiding microbial contamination, maintaining optimal environmental conditions for activity of the probiotic and optimizing the dosage and frequency of the probiotic application.
(Editor’s Note: This article was originally published in the December 2000 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Philippe Douillet, Ph.D.
Microbial EcoDynamics, Inc.
Miami, Florida, USA
[109,111,99,46,111,111,104,97,121,64,116,101,108,108,105,117,111,100,95,101,112,112,105,108,105,104,112]
Tagged With
Related Posts

Responsibility
Ammonia nitrogen dynamics in aquaculture
The major sources of ammonia in aquaculture ponds are fertilizers and feeds, and problems with high ammonia are most common in feed-based aquaculture.

Health & Welfare
Biofloc technology expanding at white shrimp farms
Biofloc technology provides high productivity, low feed-conversion ratios and a stable culture environment. Also, with viral problems and rising costs for energy, biofloc technology can help deliver sustainable production at lower cost.

Health & Welfare
Clay particles an alternative water treatment in marine fish larvae tanks
Clay particles present an alternative to water treatments with microalgae or algae paste in the culture water of marine fish larvae.

Health & Welfare
Lab evaluation methods quantify probiotic benefits
Due to the inherent complexities of field testing, definitive laboratory-based test methods can better quantify probiotic benefits.


