Experimentally cultured P. monodon fed a filamentous green alga and a microsnail show improved growth, artificial feed efficiency and profitability

The giant or black tiger shrimp (Penaeus monodon) is a prominent species of aquacultured shrimp, second only to the Pacific white shrimp (Litopenaeus vannamei) and representing about 13.4 percent of global production of farmed shrimp. It is commercially farmed in China, India, Vietnam, Indonesia and a few other Asia-Pacific countries, and also in Madagascar.
To boost productivity and feed efficiency in intensive shrimp culture, the possible use of various benthic organisms as supplementary live feeds has been explored. Various species of filamentous green and red seaweed species have been shown to enhance shrimp growth and/or survival. Although attention to profitability is necessary to consider the potential implementation of innovative techniques in intensive shrimp ponds, this is not often discussed in previous research.
The filamentous green alga, Chaetomorpha sp. and the microsnail, Stenothyra sp. are benthic organisms with no market value as fishery products but are considered promising candidates as supplementary feeds for farmed species of shrimp. Both species have been shown to promote shrimp growth, thrive in a wide range of shrimp pond conditions, particularly in terms of salinity and temperature, and grow abundantly in stagnant waters and/or channels in mangrove areas along tropical coasts.
This article – adapted and summarized from the original publication [Tsutsui I. et al. 2020. Use of a filamentous green alga (Chaetomorpha sp.) and microsnail (Stenothyra sp.) as feed at an early stage of intensive aquaculture promotes growth performance, artificial feed efficiency, and profitability of giant tiger prawn (Penaeus monodon)] – developed a simple and low-cost method to boost the productivity, feed efficiency and profitability of black or giant tiger shrimp (Penaeus monodon) aquaculture. The technique involves the culture together of filamentous green alga (Chaetomorpha sp.) and microsnail (Stenothyra sp.). to be supplementary live feeds for the shrimp and supports design innovations for commercial-scale, earthen, intensive ponds.
Study setup
This study was carried out at an outdoor experimental facility at the Shrimp Co-culture Research Laboratory (SCORL), King Mongkut’s Institute of Technology Ladkrabang (KMITL), Bangkok, Thailand. A total of 20,000 P. monodon postlarvae (PL12) were purchased from a commercial nursery in Chon Buri Province, Thailand. Following transportation to KMITL, the postlarvae (PLs) were acclimatized to the outdoor water temperature in concrete experimental ponds for approximately 30 minutes before release.
Filamentous green algae Chaetomorpha sp. and microsnail Stenothyra sp. were collected at an abandoned brackish pond in Samut Sakhon Province, Thailand, over a period of approximately three to four hours. After transportation to KMITL, the seaweed and snails were rinsed with fresh water to remove and kill any periphyton [assemblage of cyanobacteria, heterotrophic microbes, algae and detritus that attaches to submerged surfaces in aquatic ecosystems].
Six concrete ponds (9 m long × 9 m wide × 2 m deep) were used, arranged in two lines and three rows in the experimental area at KMITL (Fig. 1). The water depth was adjusted to approximately 1.2 meters in each pond, and aeration was uniformly provided for 24 hours using perforated pipes and a commercial blower. The ponds were designated as either “artificial feed only” (control) or “artificial feed and benthos” (experimental treatment), with three replicates per treatment.
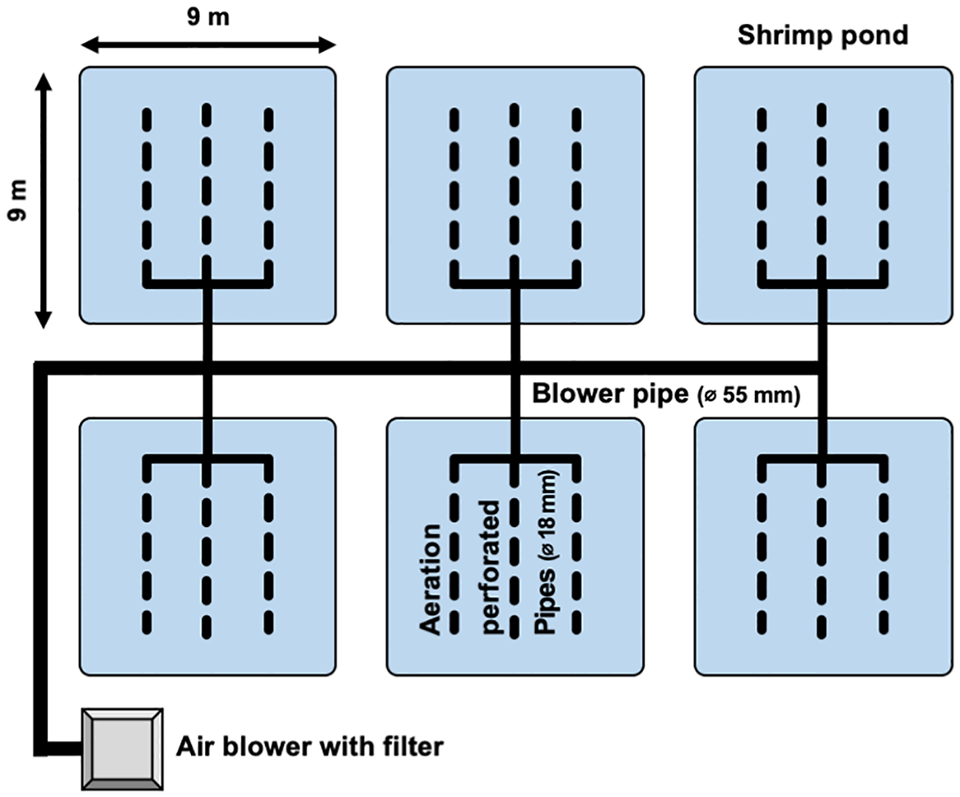
A total of 2,700 PLs were released into each pond at an initial density of approximately 33 individuals per square meter (~ 28 individuals per cubic meter). Additionally, Chaetomorpha sp. and Stenothyra sp. with mean fresh weights of 6.81 kg and 1.96 kg, respectively, were released into the experimental treatment ponds. Any other benthic organisms were removed by hand, siphon, or filtration during snorkeling observations when possible during the experimental period. All shrimp were fed commercial pelleted feed three times per day, and rations adjusted using feed trays.
For detailed information on the experimental design, measurements of monthly shrimp growth, production, benthos biomass change and apparent benthos consumption, and water quality; and statistical analyses, refer to the original publication.
Results and discussion
The individual shrimp weight at week 4 was significantly higher in the experimental treatment ponds (median, 1.12 grams; mean, 1.44 grams) than that in the control ponds (median, 0.70 grams; mean, 0.80 grams) (Fig. 2). Similarly, the mean monthly specific growth rate for weeks 0 to 4 was also significantly higher in the experimental group (23.4 percent per day) than that in the control group (21.4 percent per day) (Fig. 3). After the fourth week of culture, the shrimp in the two groups continued to exhibit significant differences in weight but no longer differed in their mean specific growth rates.
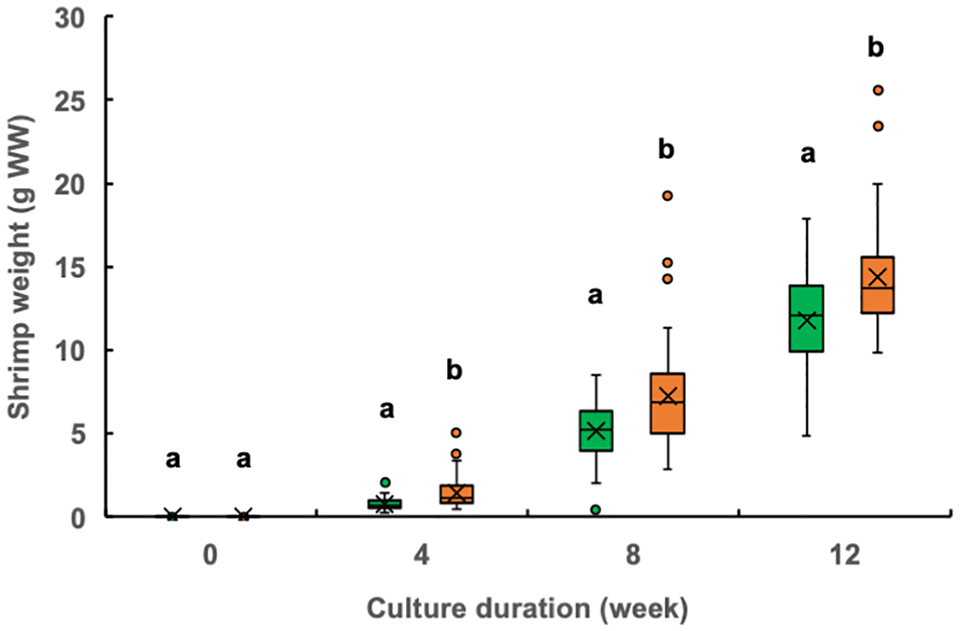
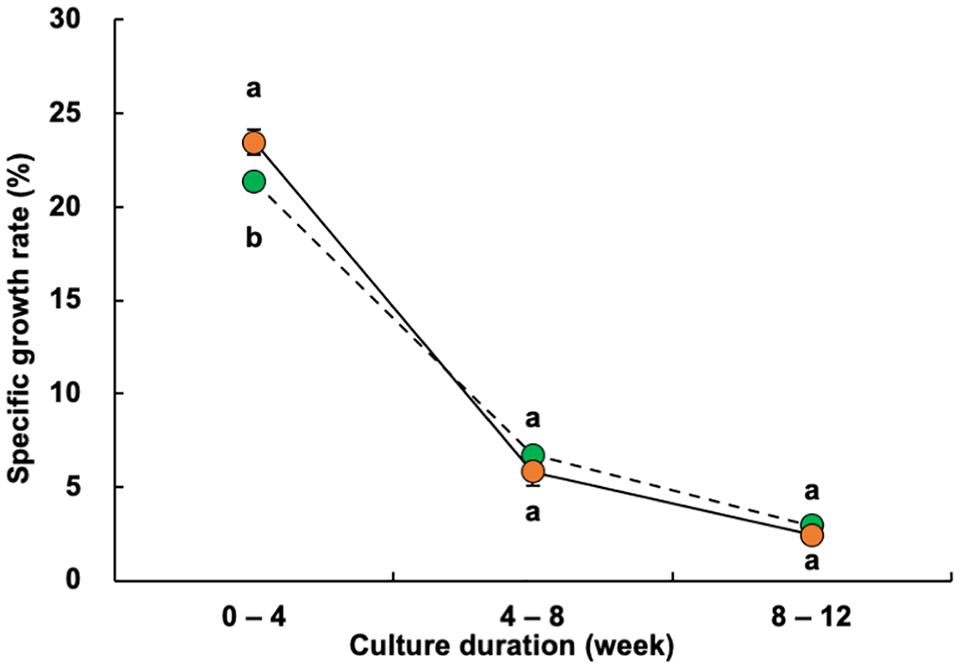
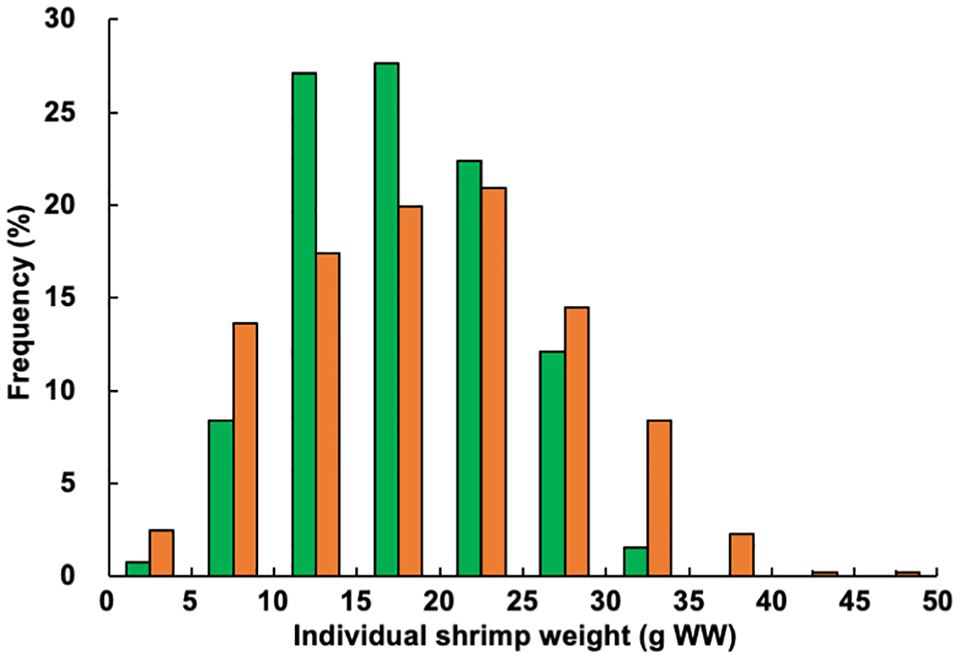
The final mean weight of individual shrimp and specific growth rate at week 15 in the experimental treatment ponds were 113.4 and 103.7 percent, respectively, of the values obtained in the control ponds, representing significant differences. Shrimp size variation was greater in the experimental treatment ponds, with a higher number of larger shrimp observed (Fig. 4). Both the mean total number of shrimp produced and survival rate were 117.5 percent of the values recorded in the control ponds, while the mean total shrimp production was 133.1 percent of the values found in the control ponds; between-treatment differences were significant for each parameter.
Various benthic organisms have been used as supplementary live feeds for cultured tropical shrimp in previous studies, and they improved shrimp growth and feed efficiencies. These studies showed that the final weights and specific growth rates of P. monodon in the experimental treatment fed benthic organisms were 115 to 157 percent and 108 to 116 percent, respectively, of the values in the control (only fed artificial feed). Final weights and specific growth rate for Pacific white shrimp (Litopenaeus vannamei) were 130 to 142 percent and 107 to 188 percent, respectively, of the control values. Similarly, the results of our study showed that P. monodon growth was significantly enhanced in the experimental group, albeit at slightly lower rates than those reported in previous studies.
Cultured shrimp growth involves higher production with higher growth at the earlier culture stages because of the exponential phase of the growth curve. Our results showed that the monthly individual shrimp weight and monthly specific growth rates were significantly higher in the experimental group than in the control group during the first four weeks of culture (Figs. 2-3). Moreover, the protein contributions of both benthic organisms provided in the experimental treatment ponds were higher in the first 4 weeks than afterwards. These results suggest that Chaetomorpha sp. and Stenothyra sp. effectively promoted shrimp growth as supplementary live feeds during the first four weeks in the early stages of growth, leading to the higher final production.
Our results showed that the artificial feed conversion ratio [aFCR; artificial feed as the only food source, excluding the contribution of other food materials, such as seaweeds] was significantly lower (89 percent) in the supplementary feed group than in the artificial feed only group. Although the aFCR we obtained was slightly higher (i.e., the feed efficiency was lower) than the values reported in previous studies (because of the smaller ratio of benthic organisms provided in this study), it is clear that the benthic organisms were efficiently utilized for shrimp production.
When considering the application of innovative techniques to real intensive shrimp ponds, it is necessary to determine their profitability such that the benefits may be predicted and extrapolated. In our study, culturing P. monodon PLs to a marketable size using the outdoor concrete experimental ponds allowed for a balanced assessment of experimental shrimp sales data and statistical analysis.
Higher sales were possible for the experimental group because of the higher mean weight of individual shrimp, total number of shrimp and total shrimp production. Although the experimental group also had higher feed costs and miscellaneous costs for benthos collection, the shrimp sales largely exceeded these costs, resulting in higher profitability.
It has previously been reported that several benthic species of seaweeds and snails enhance not only shrimp growth and feed efficiency, but also shrimp immunity, resistance to thermal stress, and the viral challenges of disease, and color when benthic species were fed as supplementary feeds. Therefore, although we only evaluated the productivity, feed efficiency and profitability of P. monodon shrimp were considered here, Chaetomorpha sp. and Stenothyra sp. may also confer these additional benefits, but more detailed studies are needed to evaluate this.
Additional effort, laborers and costs associated with the collection and propagation in ponds of benthic organisms are normally needed to operate a system with benthic organisms as supplementary feeds. However, our results demonstrated that these can be restricted to an acceptable range, making it technically feasible to apply this technique to intensive shrimp culture ponds. In the near future, we will attempt to expand this experimental culture operation on a commercial scale. To reduce benthos collection efforts and costs for farmers, as well as any impacts of shrimp culture on natural resources, future studies will examine simple techniques to increase the amounts and/or lifespans of these benthos (e.g., self-propagation in earthen ponds).
Perspectives
The productivity, feed efficiency and profitability of P. monodon shrimp were evaluated for a system in which a filamentous green alga (Chaetomorpha sp.) and a microsnail (Stenothyra sp.) were propagated together during an early culture stage and freely consumed as supplementary feeds in outdoor concrete ponds.
Approximately 44 kg of P. monodon shrimp were produced in this study from an initial weight of 5.4 grams of PLs when fed Chaetomorpha sp. (initial wet weight of 6.81 kg, or 8.37 percent to total feed consumption; estimated protein weight of 0.35 kg) and Stenothyra sp. (initial wet weight of 1.96 kg, or 2.43 percent to total feed consumption; estimated protein weight of 0.16 kg) as supplementary feeds. The productivity, feed efficiency and profitability for the group fed benthic organisms were approximately 133, 113 and 146 percent, respectively, of the values recorded for the artificial feed only group. Also, the effort and costs associated with the provision of benthic organisms could be restricted to an acceptable range.
In conclusion, the application of Chaetomorpha sp. and Stenothyra sp. as live feeds at an early stage of intensive culture improves the initial growth performance of P. monodon, and promotes shrimp productivity, feed efficiency and profitability. This innovative technique may enhance the efficiency of intensive aquaculture for P. monodon and is technically feasible using earthen culture ponds.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Isao Tsutsui
Corresponding author
Fisheries Division, Japan International Research Center for Agricultural Sciences (JIRCAS), Tsukuba, Ibaraki, Japan; and
Department of Animal Production and Fisheries, Faculty of Agricultural Technology, King Mongkut’s Institute of Technology Ladkrabang (KMITL), Bangkok, Thailand
Editor’s note: This article has 11 co-authors but only the contact information for the corresponding author is presented.
[112,106,46,111,103,46,99,114,102,102,97,64,103,110,111,99]
Tagged With
Related Posts

Health & Welfare
Biofloc technology reduces common off-flavors in channel catfish
In studies that used biofloc systems to culture channel catfish, culture tanks were susceptible to episodes of geosmin and 2-methylisoborneol and subsequent bioaccumulation of off-flavors in catfish flesh.

Health & Welfare
Managing pre-harvest off-flavors in pond-raised catfish
Although causes of pre-harvest off-flavors in catfish differ due to climate, water quality and pond hydrology, blue-green algae is a common culprit.

Aquafeeds
Use of spirulina in larviculture
Spirulina platensis is an edible planktonic filamentous bluegreen algae that can be used in fish feeds and as a supplement when feeding prawn larvae.

Responsibility
What is the role of bacteria in aquaculture ponds?
Prof. Boyd discusses the role of bacteria in the biological dynamics of ponds as essential for maintaining proper ecological function and adequate water quality.


