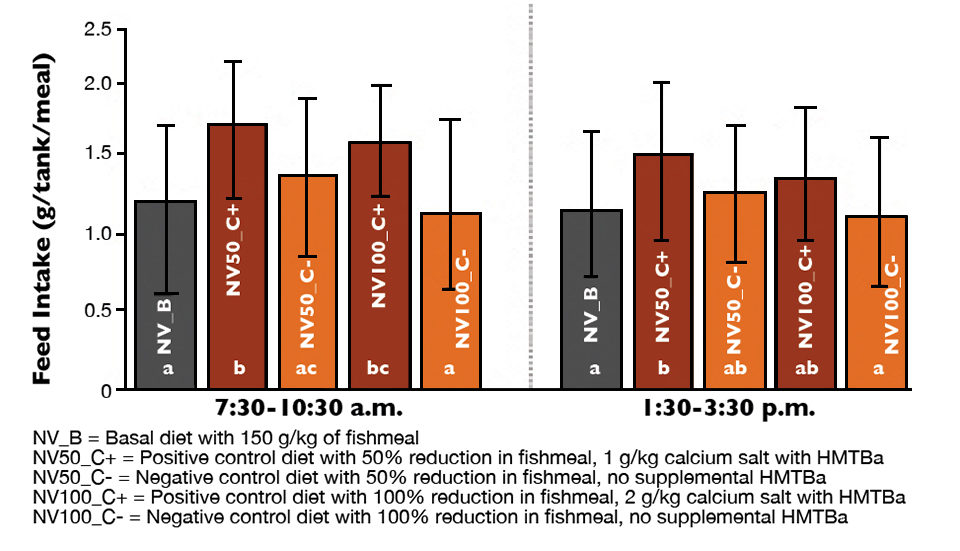
Trends toward improvements in feed intake observed for diets with reduced fishmeal and HMTBa supplementation

In shrimp diets, dietary methionine is the essential amino acid that is most impacted when fishmeal is replaced by vegetable protein sources. Methionine, one of the principal sulfur-containing amino acids, plays a critical role in the synthesis, structure and function of proteins.
In low-fishmeal diets, methionine deficiency can be avoided by supplementation with synthetic methionine. Levels as high as 8.4 grams per kg of methionine in a 35 percent protein feed are usually targeted to achieve cost-effective formulation without reducing shrimp performance.
Traditionally, crystalline DL-methionine has been used in commercial animal feeds to overcome dietary deficiencies. Another source of methionine available is 2-hydroxy-4-(methylthio)butanoic acid (HMTBa). A number of studies have reported that once ingested, HMTBa can be effectively converted to L-methionine in terrestrial and aquatic farmed species. Similarly, studies with penaeid shrimp have demonstrated that DL-methionine and HMTBa have similar efficacies for supplementing methionine in practical diets for black tiger shrimp (Penaeus monodon) and Pacific white shrimp (Litopenaeus vannamei), respectively.
The authors recently evaluated the performance of Pacific white shrimp grown at a high stocking density in a clearwater experimental rearing system. Juvenile shrimp were fed for 14 weeks with a control diet containing an intact source of methionine derived mostly from the protein of anchovy fishmeal. This control diet was compared with other diets containing progressive reductions in fishmeal in which methionine was either deficient or supplemented as HMTBa.
Experimental diets
Five experimental diets were formulated to be isonitrogenous and isoenergetic. First, a basal diet with 150.0 grams per kg of anchovy fishmeal (NV_B) and no supplemental methionine was designed. From this diet, two positive control diets (NV50_C+ and NV100_C+) were formulated to reduce fishmeal at 50 and 100 percent, respectively (Table 1). The positive controls were supplemented with MERA MetCA, a calcium salt with 84 percent HMTBa activity, at 1.0 grams per kg and 2.0 grams per kg, respectively, to target dietary methionine activity levels similar to or slightly above those achieved with the basal diet.
Nunes, Chemical composition of experimental diets, Table 1
| Ingredient (g/kg) | NV_B | NV50_C+ | NV50_C- | NV100_C+ | NV100_C- |
|---|---|---|---|---|---|
| Soybean meal | 350.0 | 457.6 | 450.0 | 487.0 | 485.2 |
| Wheat flour | 235.6 | 217.0 | 221.7 | 210.0 | 210.0 |
| Anchovy fishmeal | 150.0 | 75.0 | 75.0 | 0 | 0 |
| Poultry by-product meal | 60.0 | 60.0 | 65.7 | 60.0 | 60.0 |
| Rice, broken | 50.0 | 21.9 | 21.8 | 0.0 | 0.0 |
| Soy protein concentrate | 43.1 | 30.0 | 30.0 | 93.3 | 96.4 |
| Squid meal, whole | 0 | 20.0 | 20.0 | 20.0 | 20.0 |
| Fish oil | 15.0 | 30.0 | 30.0 | 44.0 | 44.0 |
| Soybean oil | 19.4 | 8.5 | 7.9 | 0 | 0 |
| Calcium salt with HMTBa | 0 | 1.0 | 0 | 2.0 | 0 |
| L-lysine | 0 | 0 | 0 | 0.4 | 0.3 |
| Other microingredients | 76.8 | 79.8 | 77.8 | 83.3 | 84.2 |
| Proximate Composition (g/kg, dry-matter basis) | |||||
| Moisture | 92.6 | 95.7 | 91.0 | 88.1 | 92.9 |
| Crude protein | 392.2 | 383.5 | 391.8 | 393.2 | 406.6 |
| Lipids | 70.5 | 75.9 | 70.7 | 78.0 | 60.7 |
| Total fiber | 22.7 | 26.6 | 28.3 | 27.1 | 31.7 |
| Ash | 98.7 | 97.6 | 97.9 | 95.5 | 88.4 |
| Amino Acids (g/kg, dry-matter basis) | |||||
| HMTBa | 0 | 0.65 | 0 | 1.14 | 0 |
| Methionine | 6.0 | 5.4 | 5.2 | 4.5 | 4.8 |
| Cystine | 5.4 | 5.3 | 5.4 | 5.6 | 5.7 |
| Methionine + cystine | 11.4 | 10.7 | 10.6 | 10.1 | 10.5 |
| Lysine | 19.7 | 20.4 | 18.8 | 19.4 | 22.4 |
| Cost change relative to control | – | 11.7% | 12.5% | 22.2% | 23.2% |
Two nearly equivalent diets acted as negative controls (NV50_C- and NV100_C-) without supplementation. In order to maintain levels of crude protein, total fat, gross energy, phospholipids and essential amino acids as consistently as possible across all control diets, fishmeal was replaced by soybean meal, soy protein concentrate and fish oil. Formula cost was reduced when fishmeal was progressively replaced by these ingredients (Table 1).
Feeding, water management
A total of 50, 500-L indoor tanks operating under continuous clearwater conditions were used in the study. This allowed 10 replicate tanks for each diet. Shrimp were stocked at 40 per tank (70 animals per square meter). The trial started when shrimp had reached 2.22 ± 0.19 grams wet body weight.
During the study period, animals were fed daily to satiation using feeding trays at 7 a.m. and 3 p.m. Shrimp were weighed at 72 days of rearing, then returned to their tanks to grow for an additional 24 days.
Additionally, feed attractability was measured with 4.40 ± 0.40-gram shrimp over a seven-day period in five 500-L clearwater tanks. The procedure was based on the apparent feed intake responses of the shrimp to all diets offered concurrently in the same rearing tank. Each feed was placed individually in a feeding tray rested on the tank bottom opposite each other, away from the air stones but close to the tank walls.
Water quality was kept consistent among all rearing tanks (P > 0.05). Mean water salinity, pH and temperature reached 36 ± 0.8 ppt, 7.50 ± 0.23 pH and 27.8 ± 0.51 degrees-C, respectively. Levels for nitrite and nitrate were never above the acceptable limits established for water recirculation systems. Also, un-ionized ammonia never reached lethal levels.

Results
The shrimp reached over 9 grams in 72 days and 11 grams in 96 days of rearing. Shrimp survival (92.3 ± 5.1 percent and 81.4 ± 8.0 percent), yields (461 ± 49 g/tank and 539 ± 70 grams per tank) and feed-conversion ratios (2.17 ± 0.19 and 3.12 ± 0.37) showed no significant differences among experimental treatments after 72 or 96 days of rearing (P > 0.05), respectively. On the other hand, there were statistical differences in final body weight and weekly growth.
Higher body weight was observed when shrimp were fed the basal diet or when diets were supplemented with HMTBa (Fig. 1, diets NV50_C+ and NOV100_C+). Body weights were lower when fishmeal was reduced without HMTBa supplementation. Similarly, higher weekly growth rates were found for shrimp fed either the basal diet or NV50_C+ and NOV100_C+.
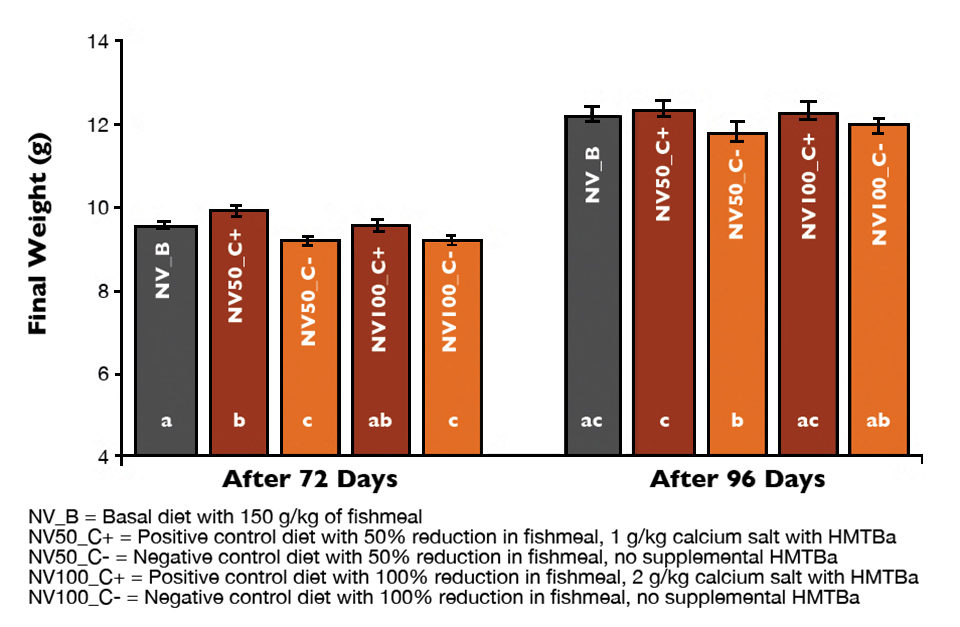
After the 14-week rearing period, growth rates averaged 0.74 grams per week with HMTBa supplementation compared to 0.70 grams per week without supplementation. A trend toward higher feed consumption was apparent at each time point for shrimp fed both supplemented feeds (Fig. 2).
Economic analysis indicated that fishmeal replacement by vegetable protein ingredients was more advantageous when HMTBa was used. Gross profit increased from 22.8 percent to as much as 41.9 percent when using diets with HMTBa.
Perspectives
Results from the study showed that L. vannamei growth, body weight, survival, yield and FCR were supported by HMTBa supplementation when 150 grams per kg of fishmeal was replaced by soybean meal at 50 and 100 percent. Trends toward improvements in feed intake were observed for diets with reduced fishmeal and HMTBa supplementation. The addition of poultry byproduct meal, squid meal and HMTBa may have helped prevent reductions in palatability typically associated with reductions in fishmeal content.
(Editor’s Note: This article was originally published in the September/October 2010 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Alberto J.P. Nunes, Ph.D.
Instituto de Ciências do Mar
Av. da Abolição, 3207 – Mereles
Fortaleza, Ceará 60165-081 Brazil[114,98,46,109,111,99,46,108,111,117,64,110,112,106,111,116,114,101,98,108,97]
-
Anant S. Bharadwaj, Ph.D.
Novus International Inc.
St. Charles, Missouri, USA -
Craig L. Browdy, Ph.D.
Novus International Inc.
Charleston, South Carolina, USA -
Jesus A. Venero, Ph.D.
Novus International Inc.
Bangkok, Thailand
Related Posts

Health & Welfare
Effect of dietary methionine on Pacific white shrimp juveniles
This study tested five diets formulated with increasing levels of methionine (Met) and Met + cysteine (Cys) and their effect on growth performance of juvenile Pacific white shrimp stocked at 50, 75 or 100 animals/m2 in a green water system.

Aquafeeds
A look at corn distillers dried grains with solubles
Corn distillers dried grains with solubles are an economical source of energy, protein and digestible phosphorus to reduce feed costs and fishmeal usage.

Health & Welfare
Amino acid supplementation reduces protein levels in pangasius diets
Trials show that supplementation with amino acids could reduce protein levels from a typical 28 percent to 23 percent in pangasius diets.

Aquafeeds
Animal byproduct concentrates useful tools in formulation
With the market volatility of fishmeal, as well as rising sustainability concerns, the aquaculture industry is seeking sources of protein, such as animal byproduct concentrates, to substitute for fishmeal.


