Monitor and manage this important water quality parameter

Total alkalinity is an important variable in water for aquaculture systems. The availability of inorganic carbon for photosynthesis tends to increase with greater alkalinity, and waters of higher alkalinity are better buffered against pH change than are lower alkalinity waters. Alkalinity is frequently measured in aquaculture systems, and especially in ponds. Managers who measure alkalinity have no doubt noted alkalinity changes of small to large amounts over time and wondered why these changes occurred.
Water in an aquaculture system may be derived from direct watershed runoff, streams, lakes, estuaries, the ocean, or wells. This water typically contains some amount of alkalinity, but if it is of low alkalinity, liming usually is done to increase alkalinity. During the production period, alkalinity may remain rather constant, increase, or decrease. Sometimes, the decrease may be drastic enough to necessitate additional liming during the culture period.
In flow-through culture systems such as raceways, water passes through quickly. There is not time for alkalinity to change greatly in response to physical, chemical, or biological processes.
Alkalinity may decline very quickly in recirculating aquaculture systems and in highly-intensive, plastic-lined culture systems without water exchange. This results from nitrification biofilters in recirculating systems and nitrification in the water column of plastic-lined ponds. Nitrification of 1 mg/L of ammonia nitrogen can reduce alkalinity by 7.14 mg/L, and a high rate of nitrification can cause a measurable loss of alkalinity within a single day.
The mere act of placing water in a pond often can cause its alkalinity to change. Suppose well water or surface runoff of low alkalinity is placed in a pond with limestone in its bottom. The limestone will dissolve and increase the alkalinity. On the other hand, if water of a moderate or high alkalinity is put in a pond with acidic bottom soil, the soil acidity will neutralize a portion of the alkalinity.

Well water from limestone-bearing aquifers may be supersaturated with carbon dioxide which resulted in dissolution of limestone and high alkalinity and calcium concentration. When exposed to the atmosphere in a pond or other culture vessel, such water loses carbon dioxide to the atmosphere, and calcium carbonate precipitates resulting in the removal of both alkalinity and calcium from the water. The precipitate is not troublesome in a pond, but it can damage eggs, fish larvae and shrimp larvae in hatcheries. Water impounded in an aquaculture system progresses towards the equilibrium alkalinity concentration as a result of equilibration with concentrations of other ions, the bottom soil, and the atmosphere.
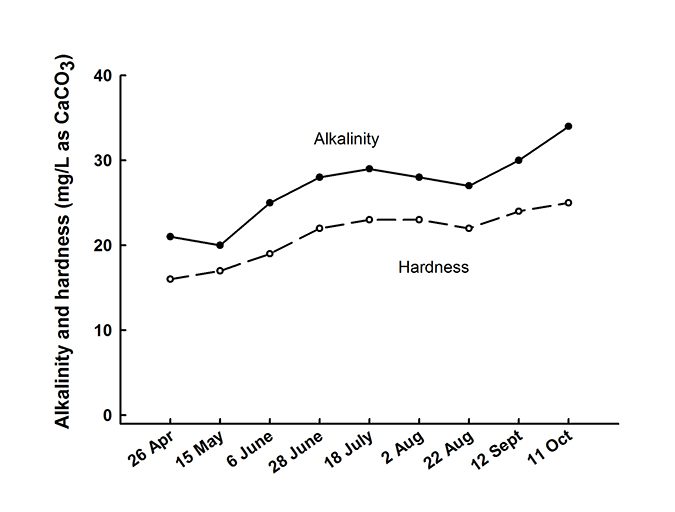
Several other processes operate in ponds to affect changes in alkalinity. During a period when evaporation exceeds precipitation, alkalinity increases. This is illustrated in Fig. 1 with data from a small pond at Auburn, Ala., USA, where evaporation typically exceeds precipitation during late spring, summer, and early fall. Of course, rainfall great enough to create overflow from ponds will lessen alkalinity, because rain usually is devoid of measurable alkalinity.
The alkalinity of estuarine water will vary seasonally depending upon the amount of freshwater inflow to the estuary. Thus, the alkalinity in coastal ponds supplied with water from estuaries depends to some extent upon alkalinity in the estuary. Of course, seawater has a rather constant alkalinity – about 120 mg/L. This favors, but does not assure, a relatively constant alkalinity in coastal ponds supplied by seawater.
Net removal from water of carbon dioxide and bicarbonate during photosynthesis by phytoplankton or other aquatic plants causes the amount of bicarbonate in water to decrease and that of carbonate to increase. This can result in supersaturation of water with calcium carbonate and result in calcium carbonate precipitation. Calcium carbonate precipitate settles slowly, and at night, accumulation of carbon dioxide in the absence of photosynthesis results in dissolution of the calcium carbonate precipitate and transformation of carbonate back to bicarbonate without loss of alkalinity. Of course, some calcium carbonate may precipitate to the bottom (known as marl formation) and an alkalinity decline can occur.
High alkalinity usually does not pose a great problem in aquaculture – this is fortunate, because there usually is no practical way of lowering alkalinity.
Aquaculture water contains nutrients that are absorbed by aquatic plants. The uptake of positive ions such as ammonium by plants must be replaced in the water by positive ions (usually H+) to maintain charge balance in the water. Thus, when plants absorb positive ions, they release hydrogen ions that depress alkalinity. The converse is true for uptake of negative ions like nitrate. Charge balance in the water is maintained by the plant releasing a negative ion (usually hydroxide), and this increases alkalinity.
Plant nutrient uptake, however, has a smaller effect on alkalinity in ponds than does nitrification. Nitrification releases two hydrogen ions for each ammonium ion oxidized to nitrate. The use of ammonium fertilizer in ponds can cause a decrease in alkalinity (Fig. 2). Feed also is a source of ammonia nitrogen to stimulate nitrification.
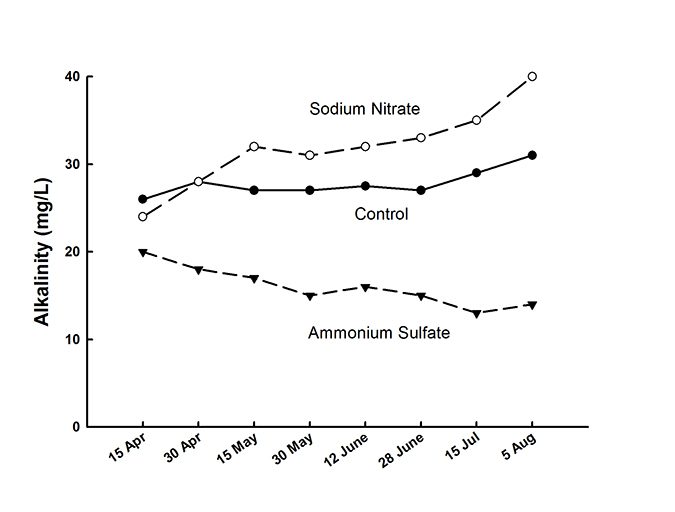
Denitrification – the process by which certain bacteria reduce nitrate to gaseous nitrogen – also occurs in ponds. Denitrification releases hydroxide ion which increases alkalinity. Alkalinity was shown to increase in ponds fertilized with sodium nitrate (Fig. 2). Denitrification releases only one hydroxide ion for each nitrate ion converted to gaseous nitrogen, while nitrification releases two hydrogen ions for each ammonium ion converted to nitrate. Even if all the nitrate resulting from nitrification is denitrified, there would still be a net loss of alkalinity.
Where water exchange rate is 5 to 10 percent or more of pond volume per day from a water source of moderate to high alkalinity, e.g., the sea, alkalinity losses are replaced continuously. In static ponds (no water exchange), alkalinity losses may be replaced by treatment with agricultural limestone or lime. There is no practical procedure for reducing high alkalinity. In water recirculating systems and in plastic-lined intensive ponds, nitrification can cause drastic losses of alkalinity in a matter of days. Traditional liming materials are not highly or quickly water soluble. The preferred method for restoring alkalinity in these systems is frequent additions of sodium bicarbonate – a readily soluble compound that increases alkalinity immediately. About 1.68 mg/L of sodium bicarbonate are necessary to restore 1 mg/L of alkalinity.

Perspectives
The upshot of this discussion is that alkalinity concentration usually changes over time in non-flow-through culture systems. Thus, alkalinity should be monitored regularly, and if it decreases to an undesirably low concentration, applications of traditional liming materials or sodium bicarbonate should be used to restore alkalinity. High alkalinity usually does not pose a great problem in aquaculture – this is fortunate, because there usually is no practical way of lowering alkalinity.
Author
-

Claude E. Boyd, Ph.D.
School of Fisheries, Aquaculture and Aquatic Sciences
Auburn University, AL 36830 USA[117,100,101,46,110,114,117,98,117,97,64,49,101,99,100,121,111,98]
Related Posts

Responsibility
Calcium and magnesium use in aquaculture
Aquatic plants and animals get the essential nutrients calcium and magnesium from water and food. Calcium concentrations impact the hydration and development of eggs in a hatchery, where calcium carbonate precipitation can be troublesome.

Responsibility
Efficiency of mechanical aeration
Although relatively expensive, mechanical aeration increases production. Asian-style paddlewheel aerators are widely used but are inefficient. Testing has led to more efficient designs that are now widely used in U.S. catfish and shrimp ponds.

Responsibility
The importance of water analysis in aquaculture
Proper monitoring of water quality in aquaculture production systems is critical to enable appropriate and timely management decisions. It requires reliable equipment, trained technicians that follow instructions and apply quality control measures, proper reagents and calibrated equipment, and appropriately collected water samples that are promptly analyzed.

Health & Welfare
The importance of iron in aquaculture systems
Iron is the fourth most abundant element in the earth’s crust, but occurs at very low concentrations in surface waters and oceans. It is an essential element for many organisms as part of many enzymes, and also has an important role in plant photosynthesis.

