Study findings highlight the urgent need for enhanced antimicrobial stewardship in the fast growing aquaculture industry

Increasing use of antimicrobials in humans and food producing animals is driving antimicrobial resistance, which is amongst this era’s defining global health challenges. Rising incidence of antimicrobial resistant pathogens of animal production significance also increase treatment failure rates, undermining sustainable food animal production and animal welfare.
Globally, rising demand for animal source nutrition is being met with a transition to increasingly intensive animal production systems. This transitional period is typically characterized by an emphasis on production volume that precedes the adoption of farm biosecurity, hygiene and management standards. In this context, non-therapeutic antimicrobial use may serve to increase growth and substitute for good animal husbandry practices. The current estimate of antimicrobial use in terrestrial food producing animals substantially exceeds human use, and is expected to grow considerably by 2030, particularly in countries with fast-growing economies like Brazil, Russia, India, China and South Africa (BRICS).
In aquaculture, production intensification and increasing incidence of aquatic animal pathogens are similarly driving antimicrobial use and antimicrobial resistance across a diversity of farmed aquatic species. Compared with antimicrobial use in terrestrial food animal production, application of antimicrobials in aquaculture provides a potentially wider environmental exposure pathway for drug distribution through water with important ecosystem health implications. Antimicrobial residues in the aquatic environment alter the environmental microbiome and, consequently, ecosystem regulatory, provisioning and supporting capacities.
Further, aquaculture settings utilizing antimicrobials may serve as reservoirs for antimicrobial resistance genes, providing routes for human and animal exposure to antimicrobial resistant bacteria. However, the levels and patterns of antimicrobial use in aquaculture globally remain largely undocumented, limiting application of targeted interventions and policies promoting sound antimicrobial stewardship in a high-growth industry.
This article – adapted and summarized from the original publication (Schar, D. et al. Global trends in antimicrobial use in aquaculture. Sci Rep 10, 21878 2020.) – presents an analysis of global antimicrobial consumption trends in aquaculture, estimating current antimicrobial use and project use to 2030 by combining species-specific antimicrobial use coefficients from a systematic review of point prevalence surveys with aquaculture production volumes. These trends are then compared with previously published data on antimicrobial use in humans and terrestrial food producing animals. The resulting estimates provide an initial foundation upon which future studies will be able to build and refine in directing iterative enhancements in antimicrobial stewardship policies.
Study setup
We estimated global trends in antimicrobial use in aquaculture in 2017 and 2030 to help target future surveillance efforts and antimicrobial stewardship policies. Baseline antimicrobial consumption and projected growth through 2030 were calculated by application of species-specific antimicrobial use coefficients to current and projected aquaculture production by species.
A systematic review of peer-review and grey literature was carried out for antimicrobial use point prevalence surveys in aquaculture between 2000 and 2019, using three primary search term categories: “antimicrobial” (antimicrobial; antibiotic; veterinary medicine); “use” (use; usage; consumption; amount; quantity); and “aquaculture” (aquaculture; aquatic; fish; shellfish; marine; freshwater).
We identified 25 studies representing 12 countries constituting 146 biomass-adjusted use rates, and from which species-specific mean antimicrobial use coefficients in milligrams per kilogram of aquatic animal biomass were obtained. Antimicrobial use coefficients by drug class were similarly obtained for analysis of use trends by class.
Antimicrobial use intensity (mg/kg) was estimated for six species groups though a systematic review of point prevalence surveys, which identified 146 species-specific antimicrobial use rates. Antimicrobial use in each country was projected by combining mean antimicrobial use coefficients per species group with OCD/FAO Agricultural Outlook and FAO FishStat production volumes.
For detailed information on the information sources and methods used in this study, refer to the original publication.
Results and discussion
At current rates, global antimicrobial consumption in aquaculture is expected to increase 33 percent between 2017 and 2030. These estimates are associated with considerable uncertainty and relatively wide uncertainty intervals due to the scarcity of point prevalence surveys on antimicrobial use in aquaculture. Global trends in antimicrobial use in aquaculture are heavily influenced by the expansion of aquaculture in Asia, and particularly in China (Fig. 1). Since 1991, China alone has accounted for more annual farmed fish output by weight than all other countries combined.
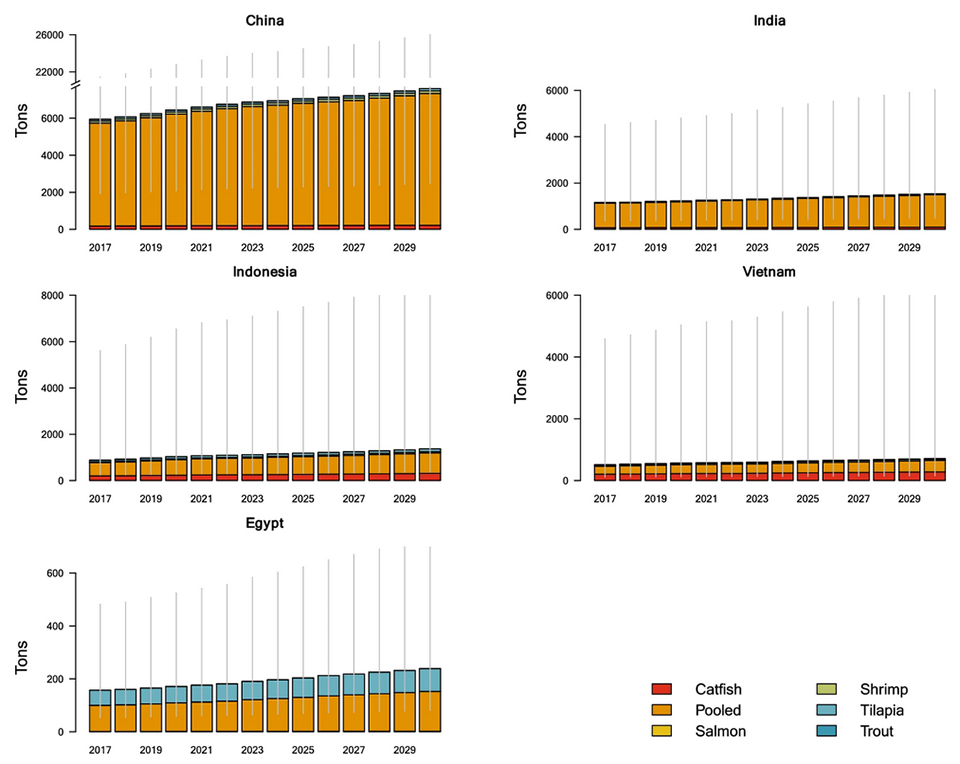
At a country-level, few estimates of antimicrobial use in aquaculture have been produced. Although this limits comparisons of our estimates with country-level use, our antimicrobial use estimates for aquaculture are within the range of a 2013 domestic consumption estimate from China, and approximately 40 percent of the low bound from a 2002 estimate in the United States.
Collectively, the five countries with the largest projected relative increase in antimicrobial consumption between 2017 and 2030 account for only 11.5 percent of global antimicrobial consumption in 2030, indicating that, with the exception of Indonesia, the countries with the fastest antimicrobial consumption growth will remain the minority contributors to global consumption projected by 2030.
By 2030, global antimicrobial use from human, terrestrial and aquatic food producing animal sectors is projected to reach 236,757 tons annually (95 percent uncertainty interval, UI 145,525 to 421,426). Proportion of use across sectors remains relatively consistent through 2030, when human use (48,608 tons), terrestrial food producing animal use (174,549 tons), and aquatic food producing animal use (13,600 tons) represent 20.5, 73.7, and 5.7 percent of global consumption, respectively.

Regarding drug classes and species, the most commonly used antimicrobial classes observed in this study – quinolones, tetracyclines, amphenicols and sulfonamides – were consistent with a previous review of antimicrobials used in the 15 highest aquaculture-producing countries. Countries with export-oriented production largely follow the regulatory structures and maximum residue limits (MRLs) established by the European Union and United States.
The four countries with the highest antimicrobial consumption identified in this study (China, India, Indonesia and Vietnam), for example, have adopted European Union MRLs to meet export requirements. Notably, enforcement of regulations remains a challenge in many countries, and parallel production systems serving domestic and export markets may encourage differing use practices.
All of the classes of antimicrobials identified in our systematic review of point prevalence surveys are classified by the World Health Organization as important for human medicine. Classes assigned to the top two classification tiers — highly important and critically important antimicrobials for human medicine — collectively represented 96 percent of all use). This finding is of particular concern given that few alternatives to these drug classes exist. It further raises the prospect of antimicrobial use in aquaculture driving resistance trends in aquatic environments, with implications for transfer of resistance genes across bacterial species. Such transfers are ecological in nature and are thus challenging to document.
Several countries have experienced dramatic reductions in antimicrobial use rates following introduction of vaccination and improved management and husbandry programs, serving as important antimicrobial stewardship models. Future strategies aimed at strengthening aquaculture production without pharmaceutical interventions may leverage advancements in bacteriophage therapy, prebiotics and probiotics, and CRISPR-Cas genome editing. Identifying solutions that can be implemented and economically self-sustaining in low- and middle-income country settings currently representing the substantial majority of aquaculture production is imperative.
Our study has some limitations. In the absence of comprehensive, standardized antimicrobial use data, our study relies upon a relatively limited collection of antimicrobial use point prevalence surveys. Despite a Chinese language search of the China National Knowledge Infrastructure (CNKI) database, surveys on antimicrobial use in China are underrepresented in our study, constituting an important knowledge gap on antimicrobial use in the world’s largest aquaculture producing nation. This limitation is particularly acute for some of the most commonly farmed fish species, such as the freshwater Cyprinidae family of species that includes carp.
Our antimicrobial consumption projections are subject to wide uncertainty intervals that likely reflect both the limited availability of surveys, from which projections were generated, and the diversity of global aquaculture production systems, practices and species. The diversity of farmed aquatic animal species greatly exceeds terrestrial food animal producing species. In 2016, 558 distinct aquatic animal species items were commercially farmed worldwide.
Although a smaller subset of 27 species groups accounted for 90 percent of global aquaculture production in 2016, by comparison, only three species groups – chickens, pigs and cattle – contributed to an equivalent level of terrestrial food animal production. Such a diversity of farmed aquatic species – compounded by polyculture production systems – presents substantial variability and challenges documentation of antimicrobial use. Currently, antimicrobial use is poorly documented even for those species of greatest production significance.
Despite these and other limitations, our estimates provide a starting point to help frame a discussion outlining near-term priorities to enhance antimicrobial use data collection.
Perspectives
Our study underscores the urgent need for standardized antimicrobial use surveillance in the aquaculture industry. Experience from countries implementing antimicrobial stewardship initiatives cite the establishment of antimicrobial use surveillance structures as the foundation for identifying risk and targeting interventions.
Robust surveillance data (1) facilitates identification of sectors and production contexts where either inappropriate use or lack of access would benefit from rebalancing; (2) enables the establishment of time-bound, measurable consumption targets and monitoring progress toward achieving these targets; and (3) when paired with resistance data, generates additional insight into the association between patterns of consumption and antimicrobial resistance trends.
A tiered approach to surveillance of antimicrobial consumption permits utilization of existing sales channel data to direct enhanced stewardship policies while structures are developed to produce iteratively more granular, farm-level consumption data. As a function of potentially higher rates of off-label use of antimicrobials in aquaculture – particularly in developing country contexts – sales data, however, may currently under-represent consumption. Labelled indications for therapeutic use in primary aquaculture species will improve attribution to – and characterization of – aquaculture use.
Our study uses current evidence to draw a first assessment of the global trends in antimicrobial use in aquaculture. Aquaculture-associated antimicrobial consumption remains a minority share of total global consumption through 2030. However, high industry growth rates, shifting dietary preferences and transitions to intensified production without corresponding management changes may drive increasing antimicrobial use in aquaculture relative to other sectors. Increases in use may be particularly significant in geographies with nascent antimicrobial consumption surveillance, regulatory and enforcement capacities.
Our findings call for urgent strengthening of surveillance for antimicrobial consumption and enhanced understanding of antimicrobial resistance transmission risk across the aquatic animal-environment-human interface, with application of targeted policies and regulatory structures promoting antimicrobial stewardship and antimicrobial efficacy as a shared global resource.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Daniel Schar
Corresponding author and Ph.D. student
Spatial Epidemiology Laboratory
Université Libre de Bruxelles, 1050, Brussels, Belgium[118,111,103,46,100,105,97,115,117,64,114,97,104,99,115,100]
-
Eili Y. Klein, Ph.D.
Center for Disease Dynamics, Economics & Policy
Washington, DC, 20005 USA -
Ramanan Laxminarayan, Ph.D.
Center for Disease Dynamics, Economics & Policy
Washington, DC, 20005, USA; and
Princeton Environmental Institute
Princeton University
Princeton, NJ, 08544 USA -
Marius Gilbert, Ph.D.
Spatial Epidemiology Laboratory
Université Libre de Bruxelles, 1050, Brussels, Belgium; and
Fonds National de la Recherche Scientifique, 1000, Brussels, Belgium -
Thomas P. Van Boeckel, Ph.D.
Center for Disease Dynamics, Economics & Policy
Washington, DC, 20005 USA; and
Institute for Environmental Decisions
ETH Zurich, 8006, Zurich, Switzerland
Tagged With
Related Posts

Intelligence
10 takeaways from GOAL 2019 in Chennai, India
The Global Aquaculture Alliance held its GOAL conference in Chennai, India, and recruited a host of experts in various fields to share their expertise.

Health & Welfare
Antimicrobial resistance: complex, unavoidable
Resistance to antibiotics is unavoidable because it is an aspect of bacterial evolution. Resistant microflora have been observed in water that has not had recent contact with antimicrobial agents.

Health & Welfare
Antibiotic-resistant bacteria, part 1
No antimicrobial agent has been developed specifically for aquaculture applications. However, some antibiotic products used to treat humans or land-based animals have been approved for use at aquaculture facilities.

Intelligence
Europeans need a new shrimp narrative
Media coverage fuels negative perceptions of farmed shrimp among Europeans. A paper – similar to one on pangasius consumption four years ago – finds the sector is doing too little to rectify ill-founded notions.


![Ad for [Aquademia]](https://www.globalseafood.org/wp-content/uploads/2025/07/aquademia_web2025_1050x125.gif)
