Water temperature, hardness and other critical factors for optimal results

The United States channel catfish (Ictalurus punctatus) industry has experienced tremendous growth during the past 40 years, becoming one of the most successful aquaculture enterprises in the country. Channel catfish surpassed other cultured foodfish to become the primary species of farm-raised fish in the United States.
Over 1 billion catfish fingerlings and fry were sold to producers in 2001, generating approximately U.S. $24 million in income for commercial hatcheries. Recent research has indicated that small improvements in current management practices can significantly improve the hatching success of channel catfish.
Hatching variability
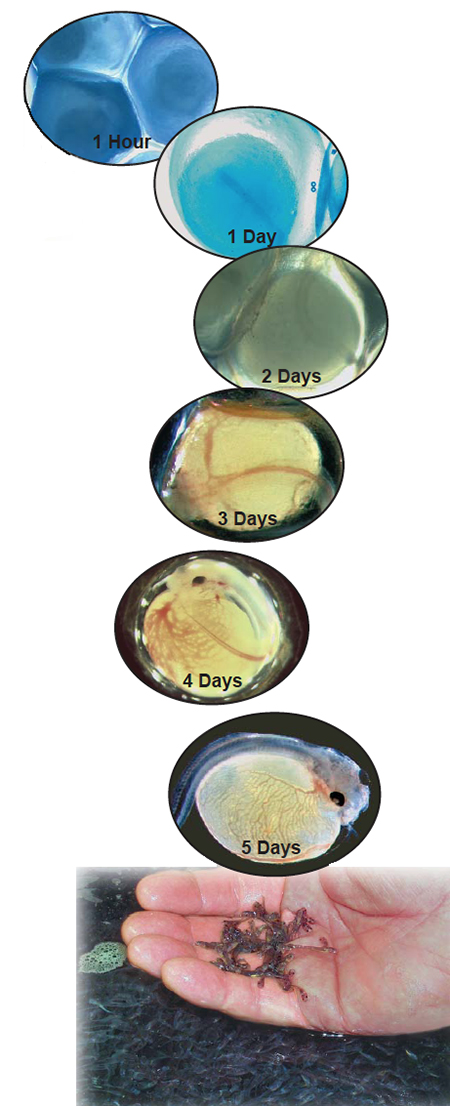
A high degree of variability associated with channel catfish hatching has been observed since the beginning of commercial catfish culture. Catfish farmers typically report average hatch rates of 60 to 70 percent. However, hatching success may actually range 0 to 100 percent within a hatchery.
The high degree of observed variability in hatching success can be attributed to many factors. Various environmental factors can be addressed through understanding the development of catfish embryos and optimizing management practices to improve environmental conditions and reduce diseases.
Incubation temperature
Temperature is an important environmental factor that affects hatching success and development. In the southern United States, channel catfish typically spawn in the spring, when water temperatures are between 21 to 29 degrees-C. At these temperatures, the embryonic period lasts five to 10 days (Fig. 1).
Effects of low temperature
In a recent study, at the Thad Cochran National Warmwater Aquaculture Center in Stoneville, Mississippi, USA looked at the effects of low incubation temperatures on catfish embryo development, hatching success, and time to hatch. Channel catfish eggs were incubated at temperatures ranging 4 to 26 degrees-C. Below 16 degrees-C, all the eggs died within 48 hours. Interestingly, survival of embryos at 16 degrees-C was dependant on the developmental stage of the embryos at the time of temperature acclimation.
Those eggs acclimated prior to the formation of the embryonic axis (approximately 36 hours post-hatch at 26 degrees-C) all died, while older egg masses survived to hatch 21 days later. These 16 degrees-C fry, however, were abnormally developed and died shortly after hatching. Fry hatched at 21 degrees-C developed normally and demonstrated post-hatch rates of growth similar to fry hatched at 26 degrees-C. However, hatching success was decreased compared to eggs from the same spawn hatched at 26 degrees-C.
Effects of high temperature
Hatchery temperatures above 26 degrees-C late in the spawning season have been suspected to cause developmental deformities such as “tripletail,” which presents itself as horizontal extensions of the caudal fin that cause fillet miscuts for catfish processors. A second study by the authors investigated the effects of temperatures up to 34 degrees-C on catfish development and hatching success.
While the results of this study clearly demonstrated a decrease in hatching success and time to hatch at higher temperatures, no correlation between tripletail and higher incubation temperatures was observed. Hatching success was found to be highest at 26 to 28 degrees-C (Fig. 2).
Water hardness
Water hardness also plays an important role in catfish embryo development. Low calcium levels in hatchery water reduce hatching success and are suspected to be another cause of skeletal deformities.
A recent study conducted in ourlaboratory investigated the effects of calcium hardness below 10 milligrams per liter during successive 24-hour stages of embryo and fry development. Eggs from a single catfish spawn were distributed into separate aquaria, with calcium supplementation turned off to three aquaria every 24 hours until the fry swam to the water surface.
Effects of low calcium
Hatching success was significantly affected by low calcium hardness. The hatching success for eggs that did not receive calcium supplementation during the first 24 hours after fertilization averaged 72 percent lower than controls which continuously received supplemental calcium (60 milligrams per liter calcium hardness as CaCO3).
Overall, hatching success was 24 percent lower for eggs that did not receive calcium prior to hatching as compared to controls. No effect of calcium on the incidence of deformities was observed. Other observations suggested physical damage during handling can increase the incidence of tripletail.
Transport delays
Generally, it is suggested that eggs not be retained at pond side for more than 30 minutes. However, eggs inevitably sit in cans or transport containers for longer periods of time.
It is assumed that long transport times and poor water conditions during transport (i.e., rising temperature and falling dissolved oxygen) result in low hatching success. In a recent study, egg masses were left in cans at pond side. At 0, 30 and 60 minutes after removal from the pond, one-third of the eggs was moved to the hatchery.
On average, water temperature in the spawning container increased 1.5 degrees-C and dissolved oxygen dropped 2 ppm over an hour. Under these conditions, a 10 to 30 percent decrease in hatching success was observed for the eggs left at pond side for an hour.
Egg treatments
Catfish eggs are commonly infected with aquatic fungi or bacteria that cause infections and poor hatching success. Dead eggs and other organic matter in culture systems provide excellent substrates for these pathogens. Currently, hatchery managers in the United States are limited to one Federal Drug Administration-approved therapeutic (formalin) and a few “low regulatory priority” compounds to control infections on catfish eggs.
Although formalin is effective, some users have safety concerns due to its suspected carcinogenicity and odoriferous nature. Two low regulatory priority compounds with excellent potential for reducing infections on catfish eggs are iodine and hydrogen peroxide. Several studies conducted in our laboratory evaluated the effect of hydrogen peroxide treatments on channel catfish hatching success when the compound was administered as a 15-minute bath or flow-through treatment.
Effects of hydrogen peroxide

Researchers compared the effectiveness of hydrogen peroxide baths and formalin baths at 1,600 milligrams per liter applied daily to eggs until eye formation to a single immersion treatment in a povidone iodine bath at a dose of 100 ppm for 10 minutes. At concentrations of 500 milligrams per liter in a daily 15-minute bath, hydrogen peroxide dissolved the membrane matrix of the eggs and caused poor survival from the premature release of the developing embryos. At half that dose, however, hatching success improved 30 percent or more when compared to eggs treated with iodine or formalin.
Hydrogen peroxide administered as a daily, 70 milligrams per liter flush treatment in flow-through hatching troughs improved hatching success more than 60 percent compared to untreated controls. Hydrogen peroxide is less expensive than formalin and provides an effective alternative for the treatment of catfish eggs that is both odor-free and noncarcinogenic.
Conclusion
While it is clear poor hatching success can be attributed to many factors, knowing the optimal conditions for handling and hatching channel catfish eggs, and following good hatchery practices will reduce the likelihood of poor hatch rates. Recommendations include:
- Maintain hatchery water temperatures at 26 to 28 degrees-C for optimal hatching success.
- Avoid transport delays from pond to hatchery. Use insulated coolers with well-oxygenated water (over 5 ppm D.O.) for transporting eggs when transport times exceed 30 minutes.
- Fill transport containers with pond water to help prevent shock due to differences in water quality and temperature.
- Avoid excessive handling of egg masses to reduce physical damage that can lead to decreased hatching success and greater potential for deformities.
- Maintain a minimum calcium hardness of 10 milligrams per liter, especially during the first 24-hour period post-fertilization.
- Treat catfish eggs daily with hydrogen peroxide baths or flush treatments until eye pigmentation is observed.
(Editor’s Note: This article was originally published in the December 2002 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Brian C. Small, Ph.D.
USDA, ARS Catfish Genetics
Research Unit
Thad Cochran National Warmwater
Aquaculture Center
141 Experiment Station Road
Stoneville, Mississippi 38776 USA[118,111,103,46,97,100,115,117,46,115,114,97,46,101,108,108,105,118,101,110,111,116,115,45,97,115,109,64,108,108,97,109,115,98]
Tagged With
Related Posts

Health & Welfare
Antigens provide immunity against ich in channel catfish trials
Vaccination against the Ich parasite is an alternative to chemical treatment. Fish develop a humoral immune response to trophont antigens, with the degree of protection related to the immunizing doses of trophonts used.

Innovation & Investment
Assessing coloration in channel catfish fillets
Because consumers look at color to gauge quality of catfish fillets, the authors developed a digital photography measurement method to assess yellowness.

Health & Welfare
Biofloc technology reduces common off-flavors in channel catfish
In studies that used biofloc systems to culture channel catfish, culture tanks were susceptible to episodes of geosmin and 2-methylisoborneol and subsequent bioaccumulation of off-flavors in catfish flesh.

Health & Welfare
Blue catfish outproduce channel catfish under low-D.O. conditions
Although there is increasing interest in blue catfish, a potential disadvantage of the fish when compared to channel catfish is their reported poorer tolerance of low dissolved-oxygen concentrations.


