Advanced technology for improved performance, genetic and environmental security
Most of the exceptional production increases in cultured shrimp production experienced during the past 20 years (from 2 percent of total world shrimp production in 1980 to more than 30 percent in 2000) were achieved using wild shrimp stocks. Now, cost-effective production increases require closed-lifecycle technology.
Domestication is necessary for improved cultivars development through genetic selection, and is essential for maintaining specific pathogen-free and specific pathogen-resistant stocks. Closed-life-cycle culture is fundamental to virtually all other mass-produced foods, including chickens, pigs, cattle, and agricultural plants.
Ploidy manipulation
The authors and co-workers are developing new technology based on ploidy manipulation in shrimp to improve culture performance and protect our valuable shrimp stocks. Ploidy refers to the number of chromosome sets carried by each cell in plants or animals. Most animals are diploid, meaning they have two copies of every chromosome. Using genetic techniques, chromosome numbers can be increased to three (triploid) or four (tetraploid) chromosome sets.
Advantages
Polyploidy, the application of triploid or tetraploid chromosomes, is widely used in cultured fish, shellfish, and plants. Usually, triploids are sterile, which can result in superior culture performance. Triploidy disrupts hormonal balances, gametogenesis, and sexual maturation. Energy devoted to sexual maturation in normal diploids can be redirected to enhanced somatic growth in triploids.
Since triploid stocks are sterile, concerns about the use of exotic species outside their normal range are eliminated. Sterility also reduces concerns about escapement of genetically distinct, domesticated stocks that might otherwise breed with wild stocks and negatively impact the population genetics of wild stocks.
Creation and maintenance of improved, domesticated shrimp stocks require large financial investments. Polyploid technology also offers a way to protect these investments.
Producing triploids
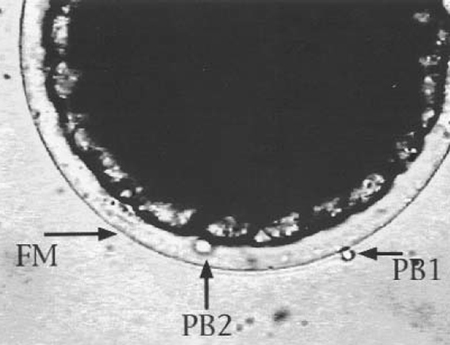
Triploids can be produced by two means. First, traumatic shock can be applied soon after egg fertilization, causing reabsorption of the second polar body into the zygote (Fig. 1). The second polar body, a product of egg meiosis, is normally extruded following egg fertilization. After polar body reabsorption, all subsequent cells of that individual are triploid with three sets of chromosomes. With this method, triploidy induction if often less than 100 percent effective.
A second method to produce triploids involves breeding normal diploids with tetraploids. This type of cross results in 100 percent triploid progeny. This method first requires that tetraploids be produced. Shocking eggs soon after fertilization produces tetraploids. But shock must be applied after the diploid chromosome set has replicated in preparation for first cell division, but before cell division begins. This disruption of first cell division results in the cell having four chromosome sets, which are maintained when the cell continues dividing.
Induced polyploidy in shrimp
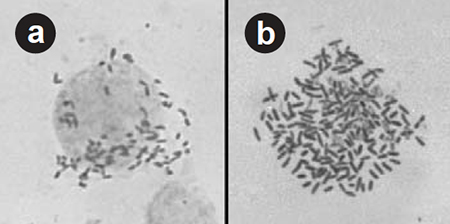
Polyploidy induction has only recently been achieved with shrimp, with most of this research occurring in China. Triploid and tetraploid fleshy shrimp (Penaeus chinensis) were produced using both temperature and chemical shocks. Chemical shocks with cytochalasin B were effective, but often produced mosaics, which is a combination of ploidy cell types within an individual.
Tetraploid Sicyonia ingesta shrimp were produced using both warm- and cold-temperature shocks, and triploid Litopenaeus vannamei have been produced using cold-temperature shocks. Arlo Fast and his colleagues successfully produced tetraploid L. vannamei using cold-temperature shocks to disrupt first mitotic cell division (Fig. 2). High Health Aquaculture in Kona, Hawaii, USA is attempting to produce polyploid P. monodon and L. vannamei, supported in part by National Science Foundation funding.
Comparing diploid and triploid shrimp
One study from China reports growth comparisons between diploid and triploid shrimp. Xiang et al. (1999) conducted growth trials with triploid P. chinensis in small, indoor tanks. Triploid shrimp were cultured for four months at three stocking densities (88, 182, and 200 shrimp per cubic meter) and their growth compared with diploid shrimp stocked at 88 shrimp per cubic meter.
At all densities, triploid shrimp grew larger than diploids, averaging 29.4, 23.8 and 1.5 percent larger for the triploids, respectively. Xiang also found that the ovaries of triploid shrimp were “undeveloped,” while no differences were observed between the testes of triploid and diploids.
Conclusion
Researchers are currently developing polyploid (P. monodon) and L. vannamei. When these shrimp are achieved, their parameters for growth, health, maturation, and other factors will be carefully evaluated against diploid performance. Technology for the mass-production of polyploids can then be established.
In the shrimp-farming future, the most advanced strains of domesticated shrimp will likely be these polyploids, with faster growth, more efficient feed conversion, and greater disease resistance. Once triploid culture performance is documented, shrimp culture based on the use of sterile postlarvae can lead to significantly improved shrimp cultivars. Genetics developers will then be able to release stocks that have been withheld until genetic security is assured.
Note: Cited references are available from the authors.
(Editor’s Note: This article was originally published in the December 2001 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Arlo W. Fast, Ph.D.
University of Hawaii at Manoa
Hawaii Institute of Marine Biology
P.O. Box 1056
Kaneohe, Hawaii 96744 USA[116,101,110,46,97,104,111,108,97,64,116,115,97,102,111,108,114,97]
-
James Wyban, Ph.D.
High Health Aquaculture
Kurtistown, Hawaii, USA
Tagged With
Related Posts

Health & Welfare
CSIRO research evaluates technologies to produce genetically protected, all-female shrimp
In Australia, CSIRO is investigating techniques to produce reproductively sterile, all-female shrimp populations through polyploidy, irradiation, or genetic engineering.

Responsibility
A helping hand to lend: UK aquaculture seeks to broaden its horizons
Aquaculture is an essential contributor to the world food security challenge, and every stakeholder has a role to play in the sector’s evolution, delegates were told at the recent Aquaculture’s Global Outlook: Embracing Internationality seminar in Edinburgh, Scotland.

Health & Welfare
Improving shellfish aquaculture through triploidy
Triploid bivalves have sparked worldwide interest since the 1980s because of their reported enhanced growth, meat content, flavor and disease resistance.

Health & Welfare
Catfish genetics and breeding
Although research on catfish genetics began in the 1960s, applications of genetic improvement in aquaculture have lagged behind other animal industries.


