TSS removal can significantly improve shrimp performance in zero-water-exchange systems

Among the various advantages of operating a zero-water-exchange rearing system is the development of a microbial community that can serve as a rich food source for farmed shrimp. However, achieving significant amounts of biofloc in the water requires strong mechanical aeration and a continuous supply of a carbon source to balance the carbon:nitrogen ratio.
These conditions can be expensive to achieve when operating with large water volumes, especially under sunlight with a high shrimp biomass. Increased availability of bioflocs also appears to be related to high water turbidity and undesirable levels of total suspended solids (TSS), which have been shown to reduce shrimp growth at high levels.
A study financed by the Brazilian Ministry of Science and Technology was carried out with juvenile Litopenaeus vannamei in a zero-water exchange system to evaluate if TSS removal could improve shrimp performance while allowing bioflocs to grow.
Rearing system, experimental design
For the study 30, 1-m3 circular outdoor tanks were not shaded from sunlight, but to keep shrimp from escaping, a net encircled the top of each tank. A floating airlift device made of polyvinyl chloride pipe was placed in the center of each rearing tank along with two air stones placed 15 cm from the tank bottom.
Fifteen tanks were equipped with a settling chamber positioned adjacent to each tank. Constructed of 60-L buckets, the chambers used air to vertically move water through a 20-mm-diameter pipe from the tank bottoms toward a chamber where solids settled. The settling chambers operated continuously throughout the rearing period and were cleaned twice weekly to remove settled material.
In this trial, a single lab-extruded diet containing 30.0 percent crude protein and 7.9 percent fat was fed to all shrimp. The diet was composed of 40.0 percent soybean meal, 29.5 percent wheat flour, 9.0 percent salmon meal, 7.0 percent dried molasses, 4.0 percent poultry by-product meal, 3.0 percent offal fishmeal, 2.0 percent soy lecithin, 2.0 percent fish oil, 1.5 percent bicalcium phosphate, 1.5 percent mineral-vitamin premix and 0.5 percent synthetic binder.
No water discharge took place during the rearing period, but fresh water was sporadically added to raise tank water levels that fell due to evaporation. Dried molasses was applied only when total ammonia nitrogen (TAN) levels reached 1.5 mg/L or above. Water was treated with sodium bicarbonate at 30 g/m3 whenever alkalinity or pH dropped below 100 mg/L calcium carbonate and 7.0, respectively.
During the rearing cycle, TAN, total alkalinity and TSS were measured every two to three weeks. Biofloc biomass was measured every two to three days in each tank with Imhoff cones. Water salinity, pH, temperature and dissolved oxygen were measured daily.
About 4,500 Litopenaeus vannamei weighing 1.97 ± 0.52 grams each were stocked in the tanks at 150 shrimp/m3 and fed daily for a 10-week period by hand broadcasting at 7:30 a.m., 11:00 a.m. and 3:30 p.m.
Results
Averaged measurements of water alkalinity (156 ± 48 mg/L), temperature (30.4 ± 0.8 degrees -C), dissolved oxygen (5.8 ± 1.1 mg/L), pH (7.92 ± 1.91) and salinity (37 ± 9 g/L) showed no statistical difference between the tanks with and without settling chambers (P > 0.05). However, both TSS and biofloc volume were statistically different (P < 0.05) between tanks with and without settling chambers.
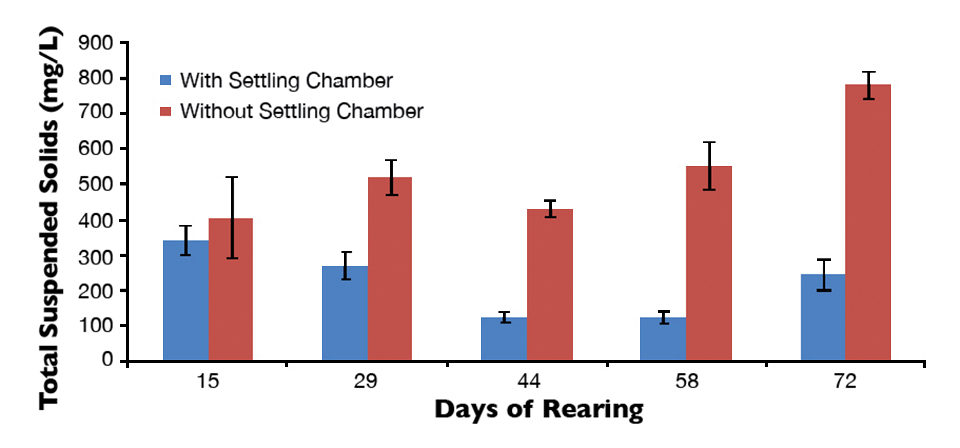
In tanks equipped with a settling chamber, TSS levels dropped over the culture period and remained below 342 ± 72 mg/L, reaching a minimum of 125 ± 27 mg/L. In tanks without a settling chamber, TSS progressively increased from a minimum of 406 ± 199 mg/L two weeks after shrimp stocking to a maximum of 783 ± 65 mg/L a day prior to shrimp harvest.
The operation of settling chambers also restricted biofloc development, as volumes were near zero in all tanks (Figure 1). This was probably the cause of higher 0.62 ± 0.71 mg/L concentrations of ammonia nitrogen throughout the rearing period in tanks equipped with settling chambers, compared to the 0.23 ± 0.24 mg/L concentrations in tanks without the chambers.
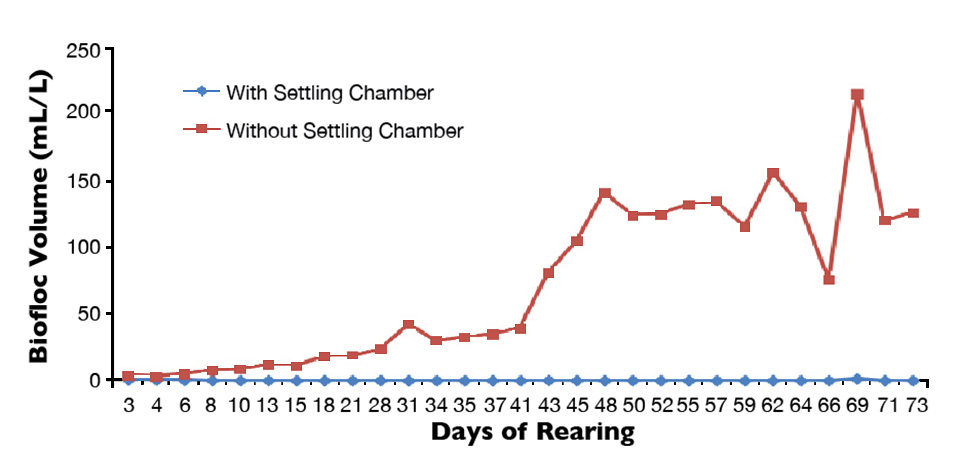
At harvest, there were significant differences in shrimp performance for the two treatments. Final shrimp survival (86.4 ± 5.75 percent vs. 79.4 ± 8.5 percent), body weight (14.49 ± 1.97 g vs. 13.62 ± 1.89 g), weekly growth (1.22 ± 0.11 g vs. 1.14 ± 0.09 g), feed-conversion ratio (1.63 ± 0.09 vs. 1.90 ± 0.21) and yield (1,877± 102 g/m3 vs. 1,622 ± 164 g/m3) were all significantly superior (P < 0.05) for tanks that operated with settling chambers throughout the rearing period.
Perspectives
Data from this study corroborated findings from other work that indicated TSS removal can significantly improve shrimp performance in zero-water-exchange systems. Under the conditions of the study, biofloc was not able to develop when settling chambers operated, which seemed to have led to higher concentrations of TAN in water.
Fine-tuning of the settling chambers to only partially remove the excess TSS considered harmful to shrimp would lessen impacts on the biofloc development in water.
(Editor’s Note: This article was originally published in the September/October 2012 print edition of the Global Aquaculture Advocate.)
Authors
-
Alberto J.P. Nunes, Ph.D.
Instituto de Ciências do Mar
Av. da Abolição, 3207 – Meireles
Fortaleza, Ceará, Brazil[114,98,46,109,111,99,46,108,111,117,64,110,112,106,111,116,114,101,98,108,97]
-
Leandro Fonseca Castro, M.S.
Instituto de Ciências do Mar
Av. da Abolição, 3207 – Meireles
Fortaleza, Ceará, Brazil
Tagged With
Related Posts

Health & Welfare
Effects of suspended solids in shrimp biofloc systems
The use of biofloc systems for shrimp culture requires monitoring of water quality parameters, particularly suspended solids levels.

Responsibility
Stocking rate effects on growing juvenile sunshine bass
A dose-response study was conducted in an outdoor biofloc system to begin quantifying the stocking rate production function for sunshine bass advanced fingerlings.

Aquafeeds
Testing shrimp growth in different biofloc systems
A study conducted with juvenile shrimp (Litopenaeus vannamei) reared in experimental biofloc systems used four treatments. Results indicate that differences in management and carbohydrate source can lead to substantial disparity in system function and production.

Innovation & Investment
Aquaculture America 2017: Communication key to the future
This year’s Aquaculture America in San Antonio, Texas, provided significant learning and networking opportunities. It successfully brought together 14 U.S. aquaculture organizations and more than 1,600 participants from Europe, Asia, Africa and Australia.


