Industry needs a wider supply of spat

The great scallop (Pecten maximus) is the target scallop species for culture in Norway. The annual production of great scallops in Norway is approximately 500 metric tons (MT), most of which is still based on harvesting wild populations.
Suitable sites for growing scallops have been found along the Norwegian coast up to 65 degrees N, and at present, about 20 shellfish farmers are using their licenses for growing scallops in intermediate or bottom culture. Final culture is based on sea-ranching, where license holders have exclusive harvesting rights for the target species within defined areas.
Growers depend on hatchery-produced spat, as previous attempts to collect spat from the wild have been unsuccessful. The spat are supplied by the only scallop hatchery in Norway, Scalpro A.S., located on the southwestern coast not far from the city of Bergen.

Variable supply
The Norwegian scallop industry needs a wider supply of large numbers of spat to succeed. During recent years, the annual production at Scalpro A.S. has been 2-3 million, 15-mm spat, but the hatchery has a much larger installed production capacity. The spat demand is increasing after being on hold due to delayed sea-ranching legislation. Large variations in growth and survival during the larval and nursery stages have been problems in ensuring a stable spat supply.
Past and present research projects at the University of Bergen, Institute of Marine Research, and Scalpro A.S. have focused on how to improve the rearing environment and enhance larval and spat yield. The larval-rearing system is very important for larval performance, and yield of ready-to-settle larvae was earlier shown to be higher in larger-scale 4,700-l rearing units than 500-l rearing units.
The introduction of flow-through instead of stagnant water in larval tanks eliminated the use of antibiotics and stabilized larval production. Nevertheless, the full effects of large-scale system conditions on metamorphosis and postlarval scallop success are not yet fully understood.
Large-scale rearing systems
Research on early scallop life stages parallels the development of better production methods and husbandry practices in the commercial hatchery. Adequate larval production results have been obtained in flow-through tanks of 3,000- to 8,000-l volume with different heights, diameters, and steepness of conical bottom.
Experimental work by the authors used 2,800-l tanks that were 2.4 m in upper height and 1.4 m in diameter with steep conical bottoms (Fig. 1). The larvae were fed a mix of cultured Tahitian Isochrysis galbana, Pavlova lutheri, Chaetoceros mulleri, and Chaetoceros calcitrans pumilum algae to meet the nutritional requirements that support normal development and optimum growth.
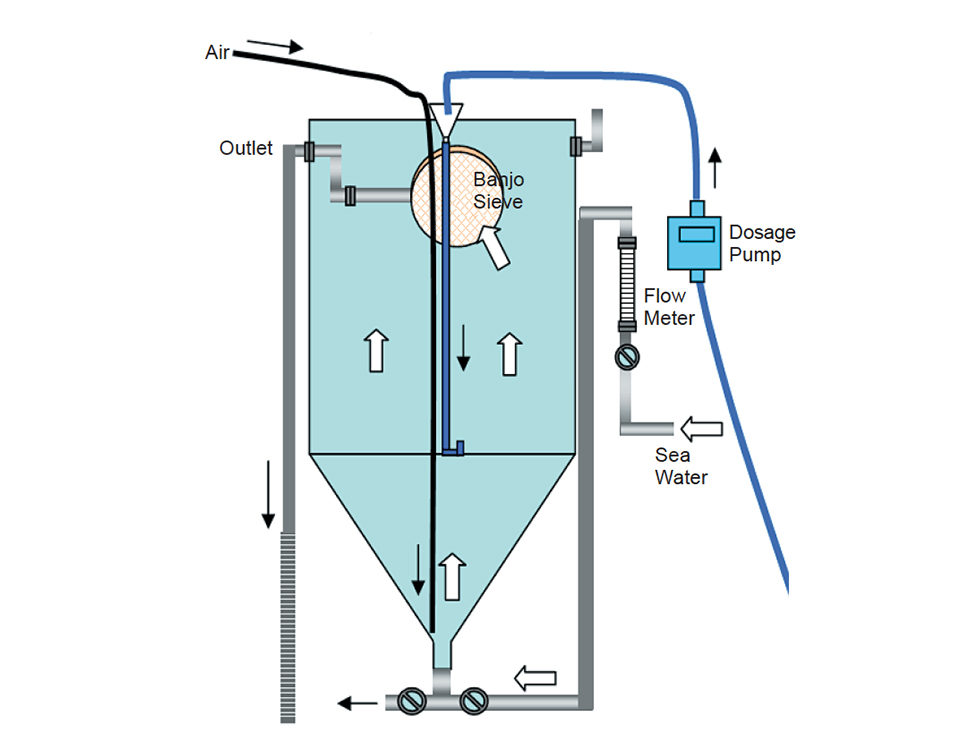
Air was added near the sea water inlet at the bottom of the tank. Banjo sieves were connected to the outlet pipe to stop larvae from escaping, with successive mesh sizes used according to the growth of the larvae. The algae were added 1.5 m from the bottom, and both larvae and algal cells were kept evenly distributed throughout the water column.
Food availability, lipid content
The intensive production of larvae demands careful management and feeding strategies that maximize growth and survival without deteriorating the water environment. Occasional observed mass mortalities of scallop larvae may have been caused by suboptimal feeding conditions related to algal concentration or larval stocking density. Both factors are vital to maintain a sustainable rearing environment, as they affect the maximum algal load that can be tolerated before overfeeding results in a build-up of organic material and decrease in water quality.
Several reports have indicated that a high lipid content in bivalve larvae is crucial for animals to successfully undergo metamorphosis and become viable postlarvae. During recent studies, the authors aimed to increase larval lipid content by facilitating increased larval food uptake, either by increasing the surrounding algal cell concentration or decreasing the larval stocking density. Feeding and sea water flow were continuous in the rearing systems, an approach considered to be the best operational method for a stable rearing environment.
Larval yields
The authors tested a series of algal concentrations (0-20 cells/µl) and initial stocking densities (1-32 larvae/ml) in six larval-rearing tanks connected to a continuous algal feeding system. Temperature was kept at about 17 degrees C.
The studies managed to increase the larval lipid content either by increasing the algal concentration or decreasing the stocking density. The authors hypothesized that the two factors significantly affected the lipid content of P. maximus larvae, and thus successful performance in the subsequent postlarval stage. However, the effects on settlement rate and early postlarval stage were minor compared to the effects on yield of competent larvae.
When initial stocking density was a constant 4 larvae/ml, the highest relative yields of both competent larvae and 4-week-old postlarvae were obtained by feeding 20 algae cells/µl. When feed concentration was kept at 16 algal cells/µl in the larval tanks, an initial stocking density of just 1 larva/ml resulted in the highest relative yield of competent larvae.
The total yield of 4-week-old postlarvae, however, was highest for the groups reared at 4 to 16 larvae/ml. By maximizing rearing conditions, the experiments showed that the maximum yield of competent larvae could be increased from 7-20 percent to 50 percent of day 3 larval number.
Future research
Further development of the scallop culture industry in Norway will depend on research and development activities at every step of the production chain. Knowledge of the early life stages is particularly important to improve spat production. Future research will focus on metamorphosis, nutritional, physiological, and genetic aspects, and grow-out techniques for spat.
Prey-predator interactions and carrying capacity are important factors for sea ranching. Health issues and the impacts of cultured scallops on the environment and wild stocks are also key, because the scallop industry will increasingly rely on the stocking of robust scallops.
Scallop farmers are required to release juveniles of local origin. Therefore, the hatchery has established conditioning regimes for broodstock from different geographical populations, and will develop stock-specific strategies for larval and spat production in the future.
(Editor’s Note: This article was originally published in the July/August 2006 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Sissel Andersen
Institute of Marine Research – Austevoll
N-5392 Storebø, Norway[111,110,46,114,109,105,64,110,101,115,114,101,100,110,97,46,108,101,115,115,105,115]
-
Dr. Gyda Christophersen
University of Bergen, Department of Biology
P. O. Box 7800, N-5020 Bergen, Norway -
Dr. Thorolf Magnesen
University of Bergen, Department of Biology
P. O. Box 7800, N-5020 Bergen, Norway
Tagged With
Related Posts

Health & Welfare
Alaska’s purple-hinge rock scallops considered for aquaculture development
Purple-hinge rock scallops are highly prized by local communities and harvested for subsistence in coastal Alaska. To evaluate the suitability of purple-hinge rock scallops for mariculture in Alaska, the authors conducted a four-year grow-out study.

Responsibility
Ailing waterways hail the oyster’s return
The Lower Hudson Estuary and Chesapeake Bay, two waterways once home to thriving oyster beds, would welcome the shellfish’s return. Aquaculture initiatives in both areas aim to reinvigorate the water and the communities they support.

Intelligence
‘Spatiotemporal patterns’ indicate improving perceptions of aquaculture
A study led by University of California Santa Barbara researchers has found that public sentiment toward aquaculture improves over time, a potentially important development with growing interest in offshore aquaculture.

Innovation & Investment
Maine scallop farmers get the hang of Japanese technique
Thanks in part to a unique “sister state” relationship that Maine shares with Aomori Prefecture, a scallop farming technique and related equipment developed in Japan are headed to the United States. Using the equipment could save growers time and money and could signal the birth of a new industry.


