Nitrifying bacteria can survive and restart the nitrification processes in substrate biofilms maintained under different conditions

Biofloc technology (BFT) is known for increased productivity in marine shrimp culture at high stocking densities, in addition to promoting improved environmental control through reduced (or zero) water exchange. The accumulation of nitrogenous compounds in the system is associated with feed application and intake by the shrimp, shrimp excretion and organic matter decomposition (e.g., uneaten feed and feces).
Undesirable levels of these toxic compounds, especially ammonia and nitrite, can induce stress and physiological changes in cultured organisms, affecting growth and survival, and may cause productivity losses. In this context, nitrifying bacteria present in the BFT system play an important role in ammonia and nitrite control, acting in their oxidation to less toxic compounds for shrimp (i.e., nitrate).
The use of artificial substrates in BFT systems is a strategy used to increase the surface area available for nitrifying bacteria fixation that colonizes the biofilm that develops throughout the culture cycle, maintaining water quality and contributing as a supplementary source of food for the animals. Given the importance of using artificial substrates to maintain water quality in intensive shrimp culture, their reuse in consecutive cycles is essential.
It is important to emphasize that this type of aerobic bacteria belongs to the so-called “adhered bacteria” that depend on a substrate for their development; once colonized, the nitrifying bacteria act in maintaining the water quality in the BFT system. In a previous study, Morais et al. observed that artificial substrates are essential for the fixation and development of ammonia-oxidizing bacteria (AOB) and nitrite-oxidizing bacteria (NOB). As a result, the reuse strategy (water or mature substrates) has been successfully used for successive cycles of super-intensive culture. The strategy of reusing inoculum with water rich in bioflocs is a common practice performed with relative success in BFT systems. The same can be used with artificial substrates, moved between tanks, or just keeping them for subsequent cycles.
Importance of aeration intensity for nitrification in biofilms in intensive biofloc systems
However, there is no consensus on how to maintain substrates in cases of extended intervals between different culture cycles, and the choice of efficient management can result in better final productivity results. Thus, it becomes important to determine what is the best management strategy that can be adopted between culture cycles in order to preserve the nitrifying bacteria community established in the biofilm.
Therefore, the aim of this study was to evaluate the effect of biofilm with different management strategies on the nitrifying bacteria community, microbial community, nitrification process efficiency and growth of Pacific white shrimp (Litopenaeus vannamei) in a biofloc system.
The authors are grateful for the financial support provided by the National Council for Scientific and Technological Development (CNPq), Research Support Foundation of the Rio Grande do Sul State (FAPERGS) and the Coordination for the Improvement of Higher Level Personnel (CAPES). Special thanks to our sponsors, All-aqua, GUABI Animal Health and Nutrition S.A., AQUATEC and TREVISAN Aerators.
Establishing nitrifying bacteria in super-intensive biofloc shrimp production
Study setup
The experiment was carried out in polyethylene units with an effective volume of 200 liters for 60 days, and it was divided into phase I and phase II, each 30 days long, respectively. A commercial, non-floating artificial substrate (Needlona®) was used in a proportion of 200 percent of the lateral area of the tank, to allow biofilm growth. The study was conducted without water exchanges.
The substrates were previously colonized within an L. vannamei BFT production system and then randomly distributed in the experimental units. For this, four treatments with four replicates were established for each management strategy used. Water samples (20 ml) and biofilm (5 cm) for microorganism analysis were collected once per week and fixed in 4 percent formalin.
Phase I
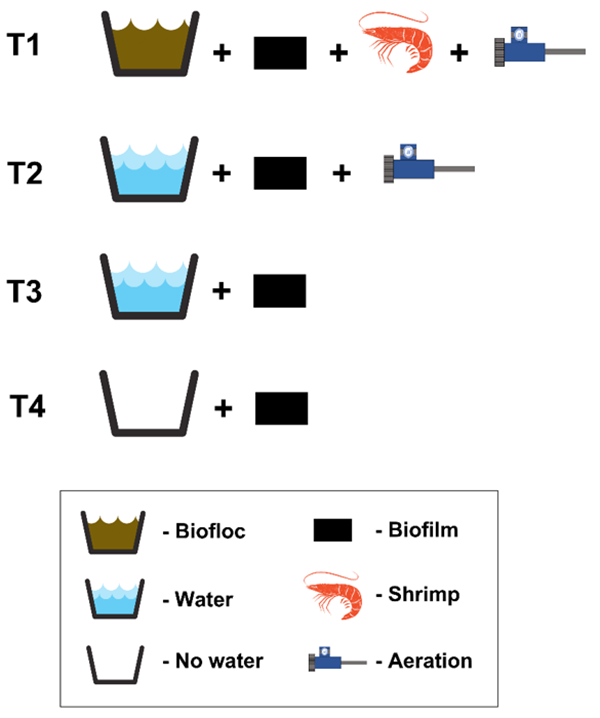
In phase I, the following treatments were established: T1 – BFT: control – biofilm + water + shrimp + aeration; T2: biofilm + water + aeration; T3: biofilm + water and T4: only biofilm (Fig. 1). The experimental units for phase I (only treatment T1 – control) were stocked with L. vannamei at a density of 500 shrimp per square meter, with a mean animal weight of 8 grams. Shrimp were fed twice per day, using a commercial, 35 percent crude protein feed (Guabi®).
Phase II

In phase II, a shrimp nursery phase (30 days) was carried out to evaluate the efficiency of the reused biofilm from the different managements implemented in phase I. Experimental units containing the matured artificial substrate were filled with filtered, chlorinated, and dechlorinated seawater and a nursery cycle carried out.
To promote biofloc growth, the carbon-nitrogen (C:N) ratio was adjusted to 15:1 with the addition of molasses when total ammonia nitrogen (TAN) concentration reached 1 mg/L. In this phase II, the experimental units were stocked with L. vannamei post-larvae at a density of 1,750 shrimp per cubic meter with an initial mean weight of 0.095 ± 0.051 grams. Shrimp were fed three times a day using a commercial 40 percent crude protein feed (Guabi®).
In both phases in this study, water quality variables (temperature, oxygen, and pH), toxic nitrogen compounds (ammonia, nitrite, and nitrate) and phosphate were monitored.
Results and discussion
Phase I
Data on the concentrations of toxic nitrogenous compounds determined during phase I are presented in Table 1. The concentrations of ammonia, nitrite and phosphate were similar among treatments. However, nitrate was higher in the T1 (control) treatment, and this major difference was due to the fact that in this treatment a mature film was used with the nitrifying bacteria (AOB and NOB) fixed and in the final stage of the nitrification process.
Ramiro, Nitrifying bacteria, Table 1
| Variables | Treatments | |||
|---|---|---|---|---|
| T1 | T2 | T3 | T4* | |
| TAN (mg/L) | 0.19 ± 0.08 | 0.12 ± 0.12 | 0.2 ± 0.18 | - |
| Nitrite (mg/L) | 0.10 ± 0.06 | 0.09 ± 0.11 | 0.08 ± 0.10 | - |
| Nitrate (mg/L) | 126.33 ± 45.62 | 9.03 ± 3.47 | 5.25 ± 3.00 | - |
| Phosphate (mg/L) | 4.17 ± 1.49 | 2.00 ± 1.50 | 1.23 ± 0.64 | - |
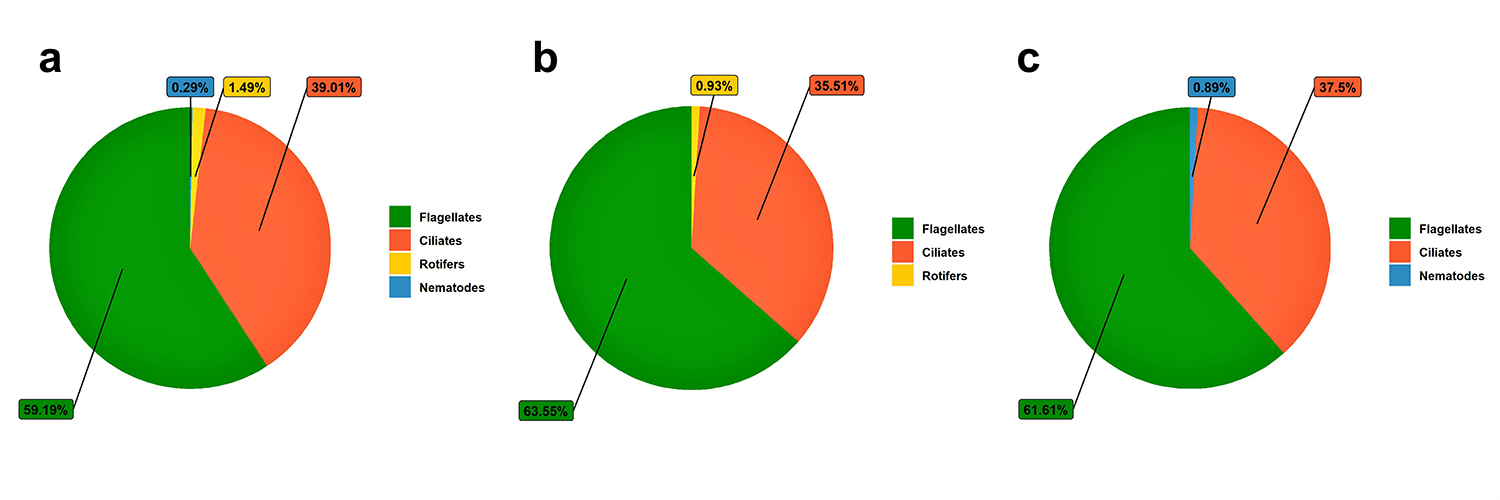
In phase I, the main water quality variables were within the ranges recommended for L. vannamei, even in the treatment without shrimp. Regarding the microorganisms in the water, we observed a marked reduction in the concentration in the treatments without shrimp and aeration, except in the control treatment. In the T2 treatment, no nematodes were found; and in the T3 treatment, no rotifers were found. The absence and reductions in concentrations of some groups of microorganisms can be explained by the adverse conditions in which the biofilm was used in this phase of the experiment. Fig. 3 shows the relative abundance of microorganisms in phase I after 30 days.
Phase II
Ammonia concentration showed a similar pattern among treatments, except for treatment T1 (control; Fig. 4a). In treatments T2, T3 and T4 there was an increase in ammonia levels in the first days of the trial, but it was under from day 7 day of culture (Fig. 4a). These results indicate the efficiency of ammonia-oxidizing bacteria after the different managements adopted for the biofilm. Nitrite (Fig. 4b), phosphate and nitrate concentrations in phase II showed a similar pattern among treatments.
In treatments T2 and T3, the concentration of nitrite was controlled from day 10 of the trial. In the T4 treatment, where the biofilm was kept out of the water, nitrite control started from day 14 of the experimental time, and levels became stable from day 18 of culture (Fig. 4b). Other water quality variables remained within the recommended range for L. vannamei. It is important to note that the study was conducted without water exchange.
In phase II, the abundance of microorganisms increased in treatments that had no shrimp and aeration during phase I. In phase II, we observed the presence of rotifers and nematodes in treatments T2, T3 and T4, which were not observed in phase I. Regardless of the management adopted, the populations of microorganisms were re-established in the water. This was probably due to the formation of a nutrient-rich medium for the re-establishment of the bacterial community, and consequently, the development of the microbial loop in the system. Fig. 5 shows the relative abundance of protozoan microorganisms in phase II of the experiment.
Regarding shrimp growth, it was similar among treatments (Fig. 6). These results show that different biofilm managements do not affect shrimp growth and survival in intensive nurseries using biofloc systems, as we found no significant differences.
Conclusion
Our bacterial analyses are in progress (even samples exposed only to air for 30 days), but based on the preliminary findings of our study, we can conclude that nitrifying bacteria can survive and restart the nitrification processes in the biofilm under different conditions.
The different management strategies we used showed that it is possible to maintain a microbial community present in the artificial substrates, after a period of absence of shrimp, aeration and even water. Furthermore, the different strategies we used in our study did not negatively affect shrimp growth.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Bianca de Oliveira Ramiro, M.S.
Laboratory of Ecology of Microorganisms Applied to Aquaculture, Institute of Oceanography, Federal University of Rio Grande, Rio Grande, Brazil
-
Wilson Wasielesky Jr., Ph.D.
Laboratory of Carcinoculture, Institute of Oceanography, Federal University of Rio Grande, Rio Grande, Brazil
-
Otávio Augusto Lacerda Ferreira Pimentel, M.S.
Laboratory of Ecology of Microorganisms Applied to Aquaculture, Institute of Oceanography, Federal University of Rio Grande, Rio Grande, Brazil
-
Natalia Pereira da Silva, B.S.E.
Laboratory of Ecology of Microorganisms Applied to Aquaculture, Institute of Oceanography, Federal University of Rio Grande, Rio Grande, Brazil
-
Genes Fernando Gonçalves Junior, B.S.E.
Laboratory of Carcinoculture, Institute of Oceanography, Federal University of Rio Grande, Rio Grande, Brazil
-
Lucélia do Valle Borges, Ph.D.
Laboratory of Carcinoculture, Institute of Oceanography, Federal University of Rio Grande, Rio Grande, Brazil
-
Dariano Krummenauer, Ph.D.
Corresponding author
Laboratory of Ecology of Microorganisms Applied to Aquaculture, Institute of Oceanography, Federal University of Rio Grande, Rio Grande, Brazil[109,111,99,46,108,105,97,109,103,64,107,111,110,97,105,114,97,100]


![Ad for [Aquademia]](https://www.globalseafood.org/wp-content/uploads/2025/07/aquademia_web2025_1050x125.gif)
