Effects on growth performance, digestive capacity, immune response and distal intestine integrity

Fishmeal sparing and replacement is still a major concern in aquaculture research. Alternative vegetable sources, in particular soybean meal (SBM) – with the highest protein content among plant-based ingredients – is one of the most promising alternative protein sources, and it already has been incorporated into the husbandry nutrition of several species.
Defatted-SBM has been considered a viable alternative to replace at least part of the fishmeal in marine fish feeds due to its availability and its 40to 48 percent crude protein, with a constant amino acid profile at low cost. However, increased use of SBM in the diets of some carnivorous fish has been related to the occurrence of enteritis – the non-infectious inflammation of the distal (part of the intestine – which can be reversed by eliminating dietary SBM. Additionally, SBM contains a high level of antinutritional factors for fish.
The totoaba (Totoaba macdonaldi) is a marine carnivorous species endemic to the Gulf of California in Mexico, which has significant potential for commercial aquaculture. There is limited information on nutritional studies evaluating soy products in its diet. One study investigating dietary soybean protein concentrate (SPC) reported improved growth and feed efficiency with acceptable haematological parameters in totoaba fed diets containing 30 to 60 percent of SPC, but hepatic damage was reported regardless of the SPC dietary level. Another study reported that when taurine – an important modulator of the intermediate metabolism of the liver – is present at 1 percent in SPC-based diets reduce the oxidative damage and restore the overall metabolism of the liver. And another recent study reported that SPC could be included at up to 34 percent in totoaba diets containing 1.5 percent taurine without affecting growth.
This article summarizes the original publication (Aquaculture 495 (2018) 78–89; https://doi.org/10.1016/j.aquaculture.2018.05.025) of the study, we carried out to evaluate the effect of graded levels of SBM on growth performance, digestive capacity, molecular expression, distal intestine and liver integrity in Totoaba macdonaldi juveniles that may result in enteritis with detrimental outcomes in fish of commercial size.

Study setup
Juvenile totoaba reared from eggs at the Marine Fish Culture Laboratory at the Center for Scientific Research and Higher Education of Ensenada (Centro de Investigación Científica y de Educación Superior de Ensenada, CICESE), Mexico, were used in the study. Forty-eight fish (71.7 ± 35.7 grams; mean ± SE) were randomly stocked into twelve 450-L cylindrical blue fiberglass tanks (four fish per tank) connected to a closed recirculation system composed of a compacted bead bed filter coupled with a fluidized biofilter and a heat pump with a daily water renewal of 5 percent. Water quality was monitored daily.
Four isoproteic and isolipidic diets were formulated to replace fishmeal (69 percent protein content), with protein levels at 0, 25, 50 and 75 percent, with a mixture of SPC (60 percent CP) and SBM (48 percent CP) at 1:4 ratio. Additionally, all diets included 1 gram/kg of krill oil as an attractant and 10 grams/kg of taurine.
Once dried, diets were packaged and stored at minus-20 degrees-C until used for the feeding trial. Four essential amino acids (lysine, methionine, threonine and arginine) were supplemented to reach equal levels found in the control diet (0 percent SBM). Although dietary treatments were formulated to replace FM protein at 0, 25, 50 and 75 percent with a mix of soybean meals, the actual SBM mixture content for each treatment was 0, 22, 44 and 64 percent.
Each dietary treatment was randomly assigned into triplicate experimental units. Fish were hand-fed daily to apparent satiation at 08:30, 12:00 and 16:00 hours for eight weeks. Daily, all uneaten feed was removed within an hour of feeding and dry weighed to determine the most accurate feed consumption rates possible.

For additional information on diet formulation; experimental design, animals and facilities; sampling; analytical methods; intestine and liver histology; gene expression quantification; and statistical analyses, please consult the original publication.
Results and discussion
The present work evaluated the effect on the growth performance, digestive capacity, as well as the progression of histological changes of the distal intestine and the expression levels of an immune system reference gene in juvenile totoabas fed with SBM.
At eight weeks, growth and production performance of juvenile totoaba was significantly affected by dietary SBM inclusion level (Table 1). Overall, fish fed SBM-free diets outperformed those fed SBM-based diets and increasing dietary SBM affected the Thermal Growth Coefficient (TGC), Feed Conversion Ratio (FCR), Condition Factor (CF), Protein Efficiency Ratio (PER) and Hepatosomatic Index (HIS). For example, fish fed 64 percent SBM diet resulted in significantly lower TGC (1.18), higher FCR (1.11) and lower CF (1.50) compared to those fed 0 percent SBM diet (TGC=1.57, FCR=0.82, CF=1.70). All SBM based diets yielded significantly lower PER (1.94–2.00) in comparison with SBM-free diet (2.50).
Fuentes, totoaba, Table 1
| Parameter | 0% SBM | 22% SBM | 44% SBM | 64% SBM | PSE | P Value |
|---|---|---|---|---|---|---|
| Thermal Growth Coefficient (TGC) | 1.61 ± 0.13a | 1.39 ± 0.14ab | 1.30 ± 0.05ab | 1.21 ± 0.19b | 0.06 | 0.029 |
| Feed Conversion Ratio (FCR) | 0.82 ± 0.03b | 1.03 ± 0.07a | 1.06 ± 0.09a | 1.11 ± 0.07a | 0.04 | 0.004 |
| Condition Factor (CF) | 1.70 ± 0.06a | 1.52 ± 0.09b | 1.51 ± 0.02b | 1.50 ± 0.02b | 0.03 | 0.007 |
| Protein Efficiency Ratio (PER) | 2.50 ± 0.09a | 2.00 ± 0.13b | 1.94 ± 0.18b | 1.87 ± 0.11b | 0.08 | 0.002 |
| Feed Intake (FI) | 0.87 ± 0.05 | 0.84 ± 0.04 | 0.98 ± 0.15 | 0.95 ± 0.07 | 0.03 | 0.263 |
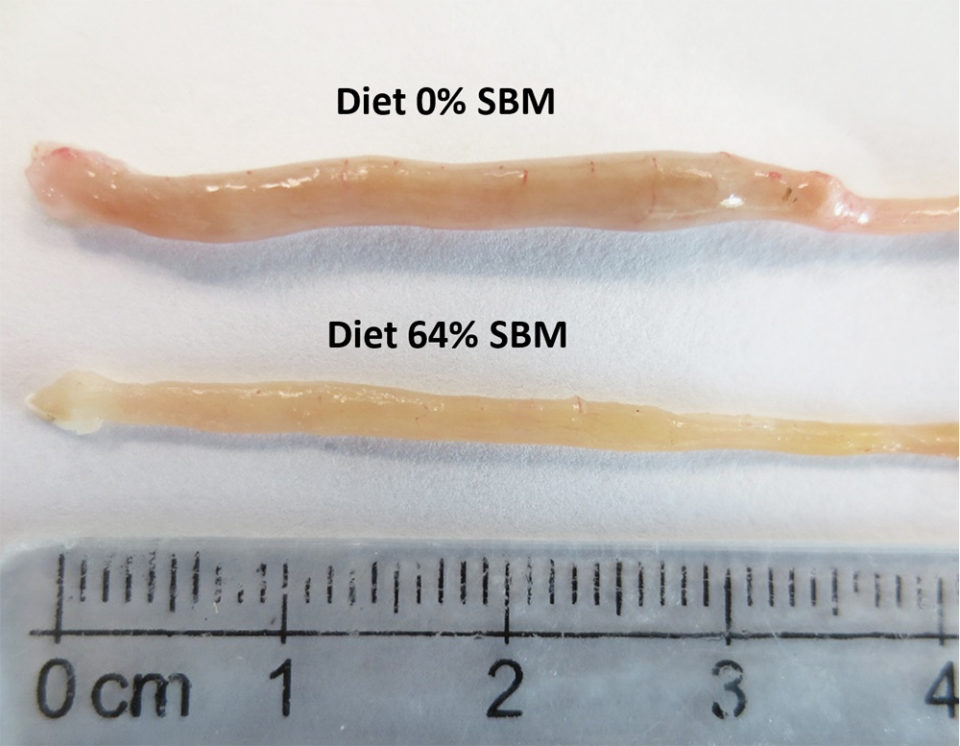
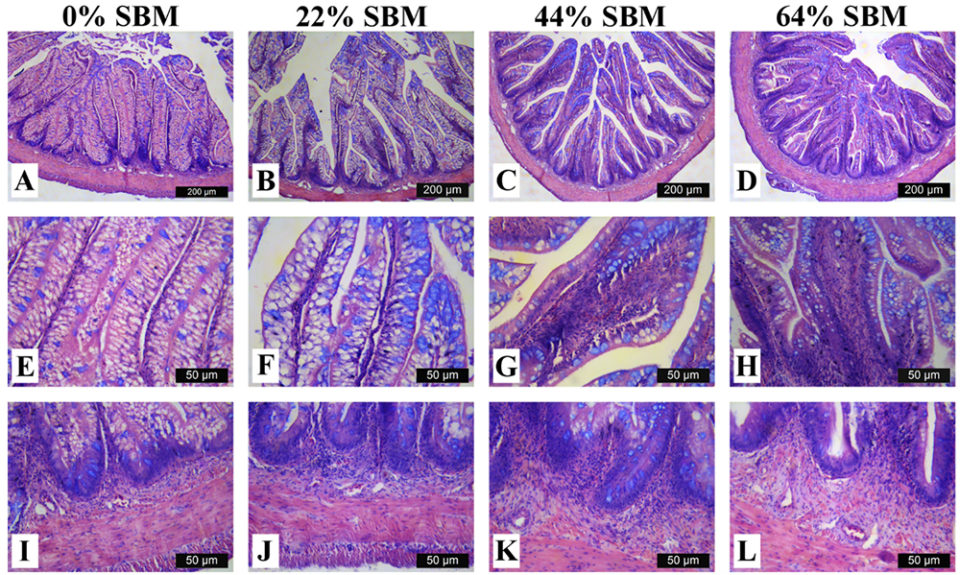
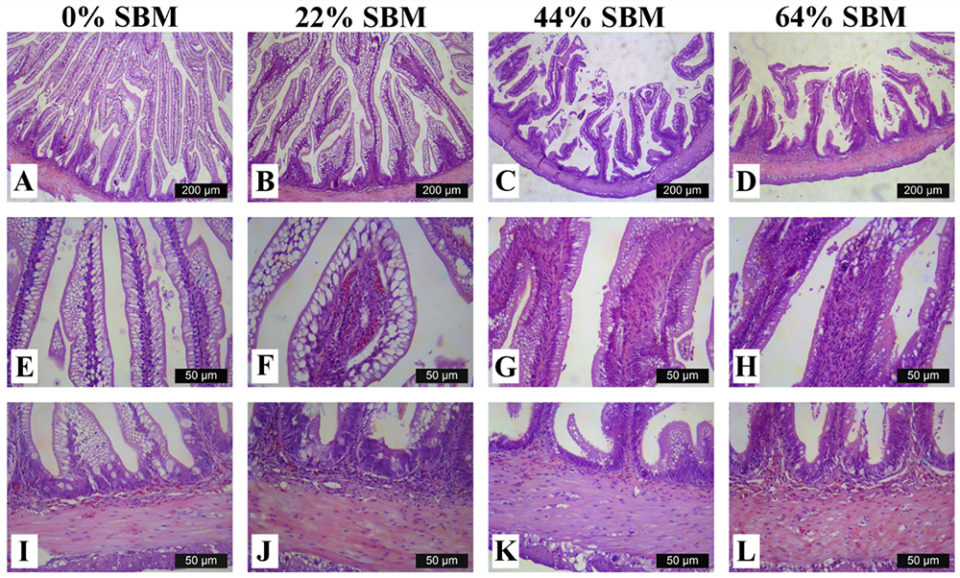
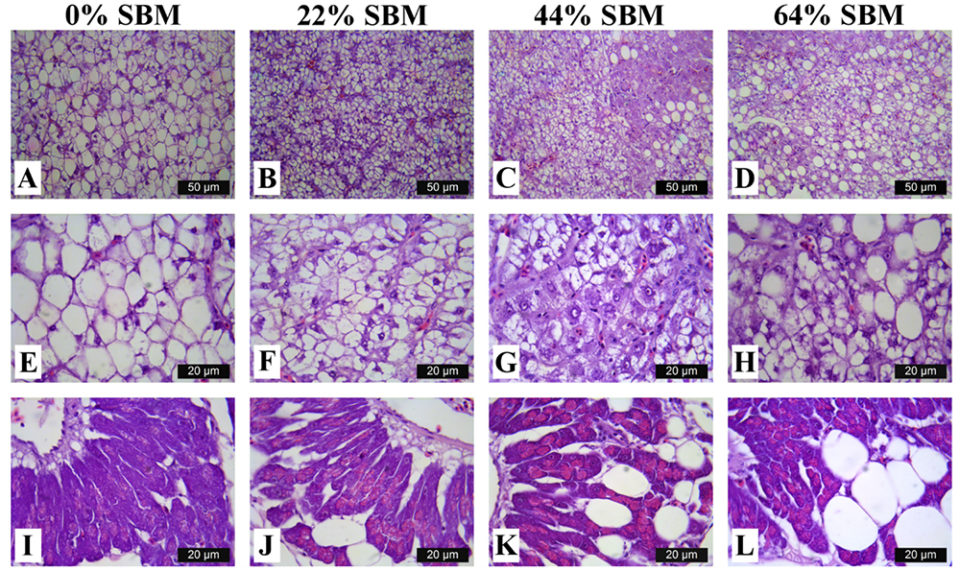
Regarding the morphology of the distal intestine and liver, experimental fish fed the control diet with 0 percent SBM, did not show any signs of morphological damage or change in the DI at four or eight weeks. But after four weeks, fish fed with SBM developed the typical characteristics associated of mucosal inflammation and intestinal damage and increased as the level of SBM increased in diet (Fig. 1). SBM decreased enterocyte height with concomitant reduction of the supranuclear vacuoles (SNV) and nuclei displacement toward apexes of enterocyte, decreased the number and length of MF and increased the coalescence of the mucosal folds (MF, Fig. 1A to D), increased the enlargement of lamina propria (LP, Fig. 1E to H) and SM (Fig. 1I to L). Few cellular infiltrations of leucocyte and eosinophilic granulocytes were also observed. Additionally, as the SBM level increased in the diet evident anatomical changes related to atrophy of the MF was observed, as well as, the hypertrophy and high hyperplasia of the GC toward the apexes of MF (Fig. 1C and D). At the end of the experiment on 8 weeks, the severity of the histological changes increased over time in fish fed with SBM (Figs. 2-3).
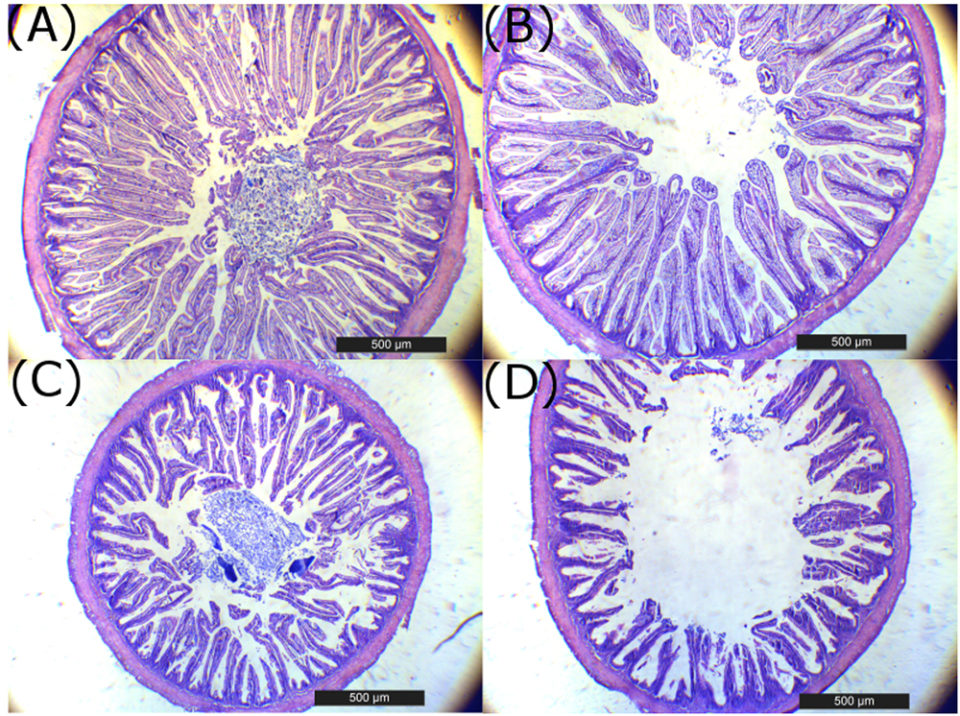
The liver histology at eight weeks in fish fed with 0 percent SBM diet showed large vacuoles of lipid with displacement of the nucleus of the hepatocytes from the central position and reduction of the sinusoid space (Fig. 4), characteristic of a fatty liver, typical of this specie. At four weeks, a significant reduction in the hepatosomatic index was observed in fish fed the 44 percent and 64 percent SBM inclusion diets. At eight weeks, the HSI tended to decrease with increasing levels of dietary SBM (HSI=1.48–0.87) for Control (0 percent SBM) and 44 percent SBM, respectively; whereas no significant differences were found for VSI and ISI at this time.
Fish performance resulted in a negative dose-dependent effect with dietary SBM inclusion. A gradually reduction in TGC is consistent with decreasing nutrient utilization and a lower PER with higher FCR. This trend was also observed in other species fed with SBM in their diets, such as cobia, silvery-black porgy, spotted rose snapper and Mediterranean yellowtail. In totoaba, this is the first work reporting the effect of SBM in the intestinal integrity assessed by histology and digestive enzyme activity. A recent study with the same species using SPC reported that diets containing up to 30 percent fishmeal replacement exhibited no adverse effects, whereas a 60 percent replacement of SPC resulted in a significantly reduced growth.
A reduction in growth performance is often associated with a lower feed intake when plant protein inclusion increases in diet. Some authors have suggested that reduce FI is due to the poor palatability of SBM caused by the bitter taste of saponins. In the present study, feed intake fails to show significant differences among dietary treatments, indicating good palatability of the diets even at the highest level of SBM. It is likely that the addition of krill oil as attractant, and the balanced supplementation of amino acids in all diets mitigate the adverse effects of palatability of the diets.
Histological evaluation of the intestines of marine fish fed vegetable ingredients helps assess in greater detail the effects on the intestinal health at the different inclusion levels, and in the overall welfare of farmed fish. The current findings demonstrate that dietary SBM, even at relatively low levels of inclusion (i.e., 22 percent), impaired not only growth and feed efficiency, but also and more importantly, the morphology and function of the digestive tract. Based on the histopathological alterations previously reported for the DI in the literature, our findings suggest that totoaba suffered enteritis that lead to a mucosal atrophy.
The same inflammatory alteration in the DI was reported for turbot, Atlantic salmon, gilthead seabream and rainbow trout. But species such as Atlantic halibut, channel catfish, European sea bass or turbot did not exhibit any inflammatory response of intestinal mucosa when fed diets with 20 to 40 percent inclusion of SBM. This suggests the totoaba has a high sensitivity to dietary SBM.
Histological analysis of totoaba liver confirms a dose-dependent damage of this organ with SBM inclusion in the diets. Totoaba is considered a moderately lean fish and have their main energy reserves in the liver, which is reflected in a liver with a high fat content. The clear reduction in fat vacuoles as SBM inclusion increased; agrees with the pattern found for the HSI. (Fig. 4A to D).
Additionally, the highest levels of SBM in the diets caused an accumulation of peripancreatic fat in the fish pancreas. We do not know for certain if the presence of high levels of peripancreatic fat, could led to a reduction in pancreas functionality. However, a reduction in trypsin, chymotrypsin and amylase activity was observed, possibly resulting in lower protein and carbohydrate digestion in the lumen of the intestine. It is possible that the lower trypsin activity could be associated to a lower enterokinase production by the enterocytes from the atrophied intestine to activate trypsinogen. Additionally, is interesting to note that the higher SBM levels in the diet the greater eosinophilyc granules in the pancreas (i.e., zymogenes), suggesting that the pancreas is compensating for the lower protease activity observed in fish fed the higher SBM diets by producing more zymogens (Fig. 4I to L).
Interleukin-8 (il-8) is one of the main immune-relevant cytokines produced during an inflammatory reaction to induce wound healing during an inflammatory process. Therefore, the relative expression level of il-8 is a good indicator of an inflammatory response. As expected, and in agreement with the literature, we observed a significant increase in il-8 expression in fish fed SBM-based diets at 4 weeks, including at the lower inclusion level (22 percent SBM), indicating a response in the posterior intestine as a result of a possible acute reaction.
Perspectives
Based on the overall performance of the fish and through the histological analysis from the distal intestine performed in the present study, we suggest the antinutritional factors in SBM are related to adverse effects found in this work, such as the reduction of the digestive enzyme activity, early inflammation reaction, tissue disruption of distal intestine, reducing nutrient absorption, late atrophy of the intestinal mucosa, with growth reduction.
In conclusion, the present study characterizes the limitations of SBM inclusion in T. macdonaldi diets, noticeably affecting the structure and physiology of distal intestine (i.e. at histological and molecular level) as well as the overall digestive enzyme activity and consequently impairing growth when included at the intermediate (44 percent SMB) and higher (64 percent SBM) levels evaluated.
Additionally, our findings demonstrated a state of intestinal atrophy in totoaba caused by the exposure of high dietary SBM inclusion levels during the time. Therefore, the present work directly suggests that SBM should be cautiously used in totoaba feeds.
References available from corresponding author.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
José Pablo Fuentes-Quesada, Dr.Sc.
CICESE, Ensenada, Baja California 22860, Mexico
-
María Teresa Viana, Dr.Sc.
Instituto de Investigaciones Oceanológicas
Universidad Autónoma de Baja California (UABC),
Km 107 Carretera Tij/Eda
22860 Ensenada, Baja California, México
-

Artur N. Rombenso, Ph.D.
Instituto de Investigaciones Oceanológicas
Universidad Autónoma de Baja California (UABC)
Km 107 carretera Tij/Eda
22860 Ensenada, Baja California, Mexico
Current Address:
Research Scientist
The Commonwealth Scientific and Industrial Research Organisation (CSIRO)
Bribie Island Research Centre
Woorim, QLD, 4507 Australia
-
Yanet Guerrero-Rentería, M.Sc
CICESE, Ensenada, Baja California 22860, México
-
Mario Nomura-Solís, M.D.
Facultad de Medicina, UABC, Ensenada, BC, México
-
Vilma Gomez-Calle, Ph.D.
Facultad de Biología
Universidad de la Serena
Coquimbo, Chile -
Juan Pablo Lazo, Ph.D.
CICESE, Ensenada, Baja California 22860, México
-
José A. Mata-Sotres, Ph.D.
Corresponding author
CONACYT, Instituto de Investigaciones Oceanológicas
UABC, México[120,109,46,117,100,101,46,99,98,97,117,64,97,116,97,109]
Tagged With
Related Posts

Responsibility
Aquaculture gives endangered totoaba a fighting chance
The tenuous fate of a pint-sized porpoise, the critically endangered vaquita, is linked to a fish targeted by poachers fueling China’s appetite for maws. The vaquita remains in peril, but aquaculture presents some hope for the totoaba.

Responsibility
Endangered totoaba has aquaculture potential in Mexico
Totoaba, a large, fast-growing fish of the Gulf of California, became endangered due to overfishing. The Mexican government therefore created an aquaculture program in Baja California to support restocking for the recovery of wild totoaba stocks.

Intelligence
A motive, and a market, for farmed fish in Mexico
Boasting ample areas for aquaculture and a robust domestic demand for seafood – not to mention its close proximity to the U.S. market – a land of opportunity lies in Mexico. Fish farming is primed to meet its potential south of the border.

Aquafeeds
Breaking the 20 percent soy barrier in fish feed
Reduced performance in fish fed high-soy feeds has been blamed on antinutrients, low methionine content and palatability issues. Pretreatment to inactivate anti-nutritional compounds and supplementation with amino acids improves soy-based feed performance, but not to control levels.



