Dietary tannic acid has potential as an antibiotic substitute for feed additives

The development of eco-friendly disease-prevention approaches is important for health management in shrimp aquaculture. Various natural plant extracts have been widely studied as new feed additives for their positive effects on growth promotion and antioxidant and immune regulation of aquatic animals.
Tannic acid (TA) is a polyphenolic compound that is ubiquitous in nature. Low-dose dietary TA has been reported as beneficial on animal growth performance, immunity, intestinal health, and antioxidative stress ability. Previous studies have indicated that the increased bacterial diversity in the intestines of organisms may play an important role in promoting nutrient digestion and absorption after a period of feeding condensed tannin-containing fish diets. Researchers have reported that low concentrations of TA can improve the health status, nutrient digestion, and growth performance of chickens and swine.
The intestine is an important organ for digestion and absorption in aquatic animals, as well as an important barrier against infectious agents and toxins. The cultivation of a healthy digestive tract has a vital impact on the health and growth of aquatic animals. In addition, intestinal microecology contains not only intestinal microbiota, but also many intestinal microbiota metabolites. Together, they affect host immune function and are important for the maintenance of intestinal homeostasis. Moreover, inflammation in the intestines of organisms further causes the proliferation of some potentially pathogenic bacteria, which may damage the barrier tissue and trigger intestinal inflammation. Tannins can reportedly regulate the abundance of beneficial or harmful bacteria in the intestine, thus altering the intestinal environment and its bacterial composition.
This article – summarized from the original publication [Gong, H. et al. 2022. Effects of Dietary Tannic Acid on Growth, Digestion, Immunity and Resistance to Ammonia Stress, and Intestinal Microbial Community in Pacific White Shrimp (Litopenaeus vannamei). Fishes 2022, 7(6), 327] –presents the results of a study to investigate the effects of dietary TA extracted from gallnut on (1) growth and feed utilization; (2) hepatopancreas and intestinal tissue structure; (3) intestinal digestive capacity and microbial community; and (4) hepatopancreas immune response of L. vannamei cultured under normal conditions and exposed to ammonia stress.
Study setup
Healthy L. vannamei (average weight 0.3 ± 0.03 g) were randomly selected from a local farming pond at Haifeng Aquaculture Co., Ltd. (Changyi, China). The shrimp were acclimated for seven days before the start of the feeding experiment. After acclimation, only shrimp in the molting stage were used. A total of 6,000 healthy shrimp were randomly distributed into four groups: TA-0, TA-200, TA-400 and TA-800. Each group had three replicate 5,000-liter plastic tanks, with each tank stocked with 500 shrimp. Each group was fed their corresponding experimental feed, and all experimental shrimp were fed to apparent satiation three times per day.
Experimental diets were formulated according to established guidelines and nutritional requirements for shrimp. A basal, commercial feed (Hengxing Group Co., Ltd., Jiaxing, China) was used as the control diet. The TA used in this study was extracted from gallnut provided by Qingdao RBT Biotechnology Co., Ltd. (Qingdao, China). Four experimental diets with different TA content were prepared: 0 mg/kg (TA-0), 200 mg/kg (TA-200), 400 mg/kg (TA-400) and 800 mg/kg (TA-800).
The feeding trial lasted for 56 days and the feeding quantity was adjusted according to the growth of the shrimp. The water quality parameters in the test were dissolved oxygen (DO) > 6 mg/L; salinity 32 ± 0.5; temperature 28 ± 0.5 degrees-C and pH 7.8–8.2. About 30 percent of the water in each water tank was replaced every day and the shrimp feces and uneaten feed were cleaned out each time.
At the end of the 56-day feeding trial, hepatopancreas samples from each tank were randomly collected for biochemical and gene expression analyses, and the corresponding intestines were sampled for digestive enzyme activity and microbial community analysis. All experimental samples were frozen and stored in liquid nitrogen for further analysis.
For detailed information on the experimental design, animal husbandry, diet preparation, and analyses performed, refer to the original article.
Results and discussion
In this study, supplementation with 400–800 mg/kg TA in diets promoted growth, reduced the feed conversion ratio, and improved the survival rate of L. vannamei after ammonia stress.
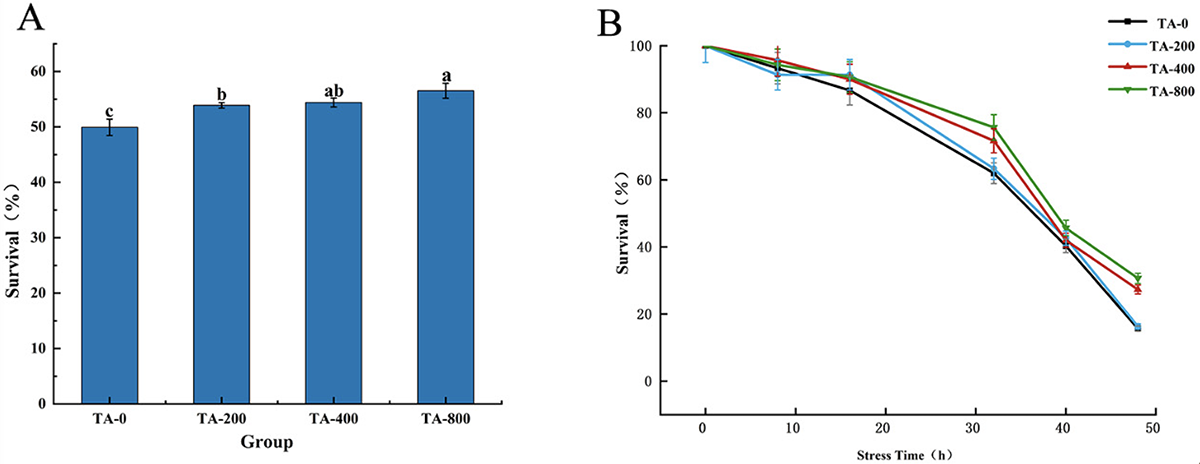
The shrimp fed the TA diet had a lower FCR and higher weight gain than those fed the TA-0 diet. The TA-400 group had the highest growth performance and feed utilization. After a 56-day feeding trial, the survival rate of the shrimp in the three TA groups was significantly higher than that in the TA-0 group, and the TN-800 group’s survival rate was significantly higher than that of the TN-200 group (Fig. 1A). Additionally, after ammonia stress for 48 hours, the survival rate of the TA-0 group was approximately 15.67 percent, and that of the TA-800 group was approximately 30.67 percent. The TA-800 group survival rate was significantly higher than that of the TA-0 group (Fig. 1B).
The activity of digestive enzymes is directly related to the digestion and absorption of nutritional material and the animal’s growth. It has been reported that a low dosage of TA has positive effects on the digestion and absorption function of organisms. In this study, dietary TA increased the activity of digestive enzymes in the intestine of L. vannamei, indicating that TA could enhance nutrient digestibility and contribute to improving growth performance.
Also, dietary TA might enhance the antioxidant capabilities and non-specific immunity of shrimp to defend against ammonia stress.
The intestinal microbiota is closely associated with host health. Our results indicate that TA supplementation for 56 days increased the diversity and altered the composition of the intestinal microbiota in L. vannamei. In our experimental results, several major bacterial genera also showed obvious differences among the experimental groups. Among them, we focused on bacteria that are relevant to host health, such as Rhodobacteracea, which can contribute to the global carbon and nitrogen cycle by breaking down large amounts of organic material.
A previous study reported these bacteria play an important role in maintaining health and promoting the growth of aquatic animals. Our data show that the highest relative abundance of Rhodobacteracea was observed in the TA-800 group, which was consistent with the best growth performance of L. vannamei. These results suggest that dietary TA can promote the growth of shrimp by regulating the intestinal flora abundance. In addition, Paracoccus sp. (Proteobacteria) is a denitrifying bacterium that can help reduce nitrate to molecular nitrogen. In our study, the abundance of Paracoccus increased in some TA groups, and TA increased the abundance of beneficial bacteria in the intestine.
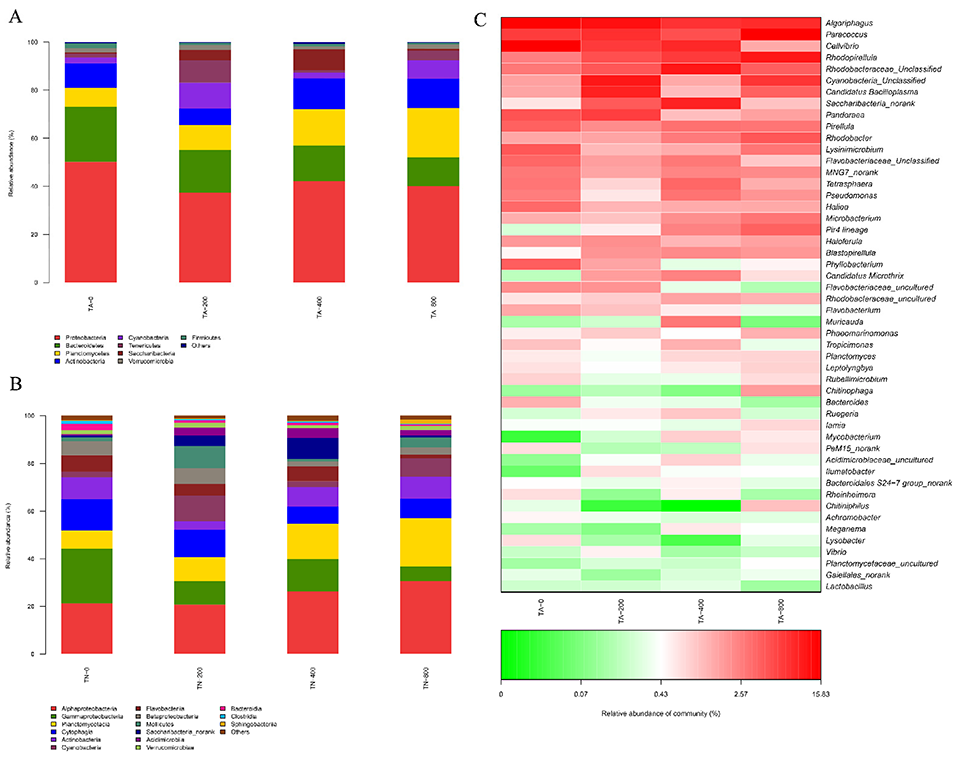
Various opportunistic pathogens can occur in the shrimp intestine, such as those in the bacterial family Flavobacteriaceae. We found that the decreased abundance of Flavobacteriaceae indicated that dietary TA might reduce the pathogen abundance in the shrimp intestine. Vibrio spp. are common pathogens in aquatic animals, and researchers have observed that dietary TA promoted antibacterial activity by reducing the content of iron ions. From our results, the reduced levels of Vibrio spp. indicated that TA reduced the risk of putative opportunistic pathogens in the L. vannamei intestine.
Overall, our data showed that dietary TA could regulate the intestinal microbiota metabolic function of L. vannamei. Therefore, dietary TA supplementation could cause intestinal microbiotic variations in L. vannamei, and the increased microbial diversity might contribute to counteracting the adverse effects of ammonia exposure.
Perspectives
Results from this study showed that dietary tannic acid improved growth, digestion, nonspecific immunity, resistance to ammonia stress and the intestinal microbial community. TA did not have any negative effects or severe changes in morphological structure of the intestine and hepatopancreas in L. vannamei.
Furthermore, TA not only enhanced the relative abundances of beneficial bacteria, but also lowered the relative abundance of opportunistic pathogenic bacteria in shrimp. The results showed that 400–800 mg/kg TA represents the most application value for shrimp aquaculture.
The interactions among growth performance, antioxidant capability and the composition of intestinal microbiota in L. vannamei deserve further exploration.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Dr. Ping Chen
Corresponding author
Key Laboratory for Development of Marine Fisheries, Ministry of Agriculture and Rural Affairs, Yellow Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences, Qingdao 266071, China[110,99,46,99,97,46,105,114,102,115,121,64,103,110,105,112,110,101,104,99]
Related Posts

Health & Welfare
Egg saprolegniasis in a commercial sunshine bass hatchery
Fungal growth on the eggs caused by the water mold Saprolegnia spp. is an obstacle to the larval production of sunshine bass. This study confirmed that copper sulfate can be an economical and valuable resource in hatchery management for the control of saprolegniasis on sunshine bass eggs in McDonald jars.

Health & Welfare
Acclimating shrimp postlarvae before pond stocking
Shrimp postlarvae acclimation before stocking into the various growout systems (ponds, raceways, tanks) is a critical – and often overlooked, sometimes taken for granted – step in the shrimp culture process. Various water quality parameters should be changed slowly so that the young shrimp have the time to gradually adapt to the new conditions.

Responsibility
Ammonia nitrogen dynamics in aquaculture
The major sources of ammonia in aquaculture ponds are fertilizers and feeds, and problems with high ammonia are most common in feed-based aquaculture.

Health & Welfare
Estimating heritability of ammonia tolerance in Pacific white shrimp
This study estimated the heritability of ammonia tolerance in Pacific white juveniles. Results suggest that rapid genetic gains could be obtained.


![Ad for [membership]](https://www.globalseafood.org/wp-content/uploads/2025/07/membership_web2025_1050x125.gif)
