Lipids play critical nutritional roles in early stages of farmed fish and shellfish

Phospholipid is a general term that includes all lipids containing phosphorus. However, it is a term often mistakenly equated with phosphoglycerides, the most common of the phospholipids. The first phospholipid identified as such in biological tissues was lecithin, or phosphatidylcholine, in the egg yolk, by Theodore Nicolas Gobley, a French chemist and pharmacist, in 1847.
Phospholipids are the major constituents of cell membranes and are vital to the normal function of every cell and organ. They maintain cell structure and function and have regulatory activities within the membranes and outside the cell.
For instance, they serve as second messengers in cell signaling, an essential process in regulating cell growth, proliferation, differentiation, metabolism, nutrient uptake, ion transport and even programmed cell death.
Phospholipids act as emulsifiers and facilitate the digestion and absorption of fatty acids, cholesterol and other lipophilic nutrients. They also have a role in the transport of lipids, not only in the transport of absorbed lipids from the gut into the hemolymph, but also the transport of lipids between tissues and organs.
Phospholipids are important precursors for a range of highly biologically active mediators of metabolism and physiology, including eicosanoids, diacylglycerol (DAG), inositol phosphates and platelet activating factors (PAFs).
Dietary phospholipids may serve as a source of choline, inositol, LC-PUFAs or even energy. For early stages of crustaceans, it has been suggested that phospholipids presented in the diet serve as a direct source of these nutrients. In fish feeds, phospholipids can account for 5 to 25 percent of the total lipids, depending on lipid content and formulation of the feed.
Sources

All products of plant and animal origin contain phospholipids, but not all contain high levels of specific phospholipids. In particular, vegetable phospholipids are deficient in long-chain polyunsaturated fatty acids (LC-PUFAs) like EPA and DHA. Soya phospholipids are the most widely used phospholipid source for animal nutrition, having a relatively low PC (phosphatidylcholine; a class of phospholipids) content and low omega-3 fatty acid levels.
Classification
Phospholipids are classified into four main classes: glycerophospholipids, sphingolipids, ether phospholipids and phonophospholipids, depending on their backbones and bonding types. In contrast to other phospholipids species, glycerophospholipids have been widely used in foods, pharmaceuticals and other industrial fields.
Digestion
The digestion of dietary phospholipids is not well studied in fish, but it is presumed that the mechanisms are generally similar to those in mammals, whereby phospholipids are digested by intestinal phospholipase A2, secreted by the pancreas, resulting in the formation of 1-acyl lyso-phospholipids and free fatty acids that are absorbed by the intestinal mucosal cells.
The lack of a discrete pancreas in most teleost species has hampered studies on intestinal lipolysis in fish. Phospholipase A2 activity was only detected several days after mouth opening in marine fish larvae.
Absorption
The mechanisms of absorption of the products of phospholipid digestion have not been extensively studied in fish, but are assumed to be generally similar to those in mammals. Thus, the hydrolytic products – 1-acyl lyso-phospholipids and free fatty acids – will be associated with all the other products of fat digestion in mixed micelles with bile salts that diffuse to the intestinal mucosa where uptake into the enterocytes occurs, probably mainly by passive diffusion.
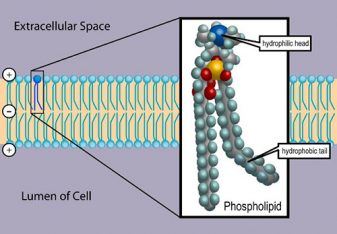
Transport
Phospholipids are transported in the blood of most fishes as lipoproteins, as they are in mammals. Lipoproteins include chylomicrons, very-low density lipoproteins (VLDL), low density lipoprotein (LDL) and high density lipoprotein (HDL) containing 8, 21, 25 and 29 percent of total weight as phospholipids, respectively. However, as a percentage of the total lipids, the proportion of phospholipids in various fishes vary widely.
Although virtually unstudied in fishes, by analogy with the system characterized in mammals, phospholipids transport in the liver and some other tissues of fish happens through receptor-mediated endocytosis or exchanged via direct interaction with the endothelial cell membranes in the tissues.
Role of phospholipids in fish and shellfish nutrition
Phospholipids are required for optimal growth, survival, prevention of skeletal deformities and, possibly, stress resistance in larval and early juvenile fishes, both marine and freshwater species. In larvae fed diets rich in triacylglycerol, lack of sufficient dietary phospholipids limits lipoprotein synthesis in enterocytes, leading to impaired transport of lipid to the tissues.
Fish larvae during embryonic development and subsequent stages in their natural feeding habitat always have different types of phospholipid classes at their disposal. Those phospholipids originate from the egg reserves and later from the live food organisms. Under artificial conditions, problems arise when larvae are offered formulated diets with inadequate quantity of phospholipids. Phospholipids are known to act as emulsifiers in the intestinal lumen. They help in the absorption of dietary neutral lipids such as cholesterol and triglycerides, as found in crustacean larvae.
The role of dietary lecithin as emulsifier is well established in seabream larvae. Larval stages are very sensitive to phospholipid deficiency. Zoea I/II stages of P. japonicus suffered 100 percent mortality before reaching the mysis stage when fed a phospholipids-deficient diet. For first-feeding carp larvae, the supplementation of 2 percent phospholipids to the diet highly improved both survival and final weight compared to that of carp fed phospholipids-free diets. Similar drastic effects on survival were shown for larvae of red seabream when fed a phospholipids-free diets.
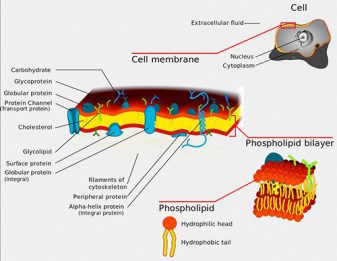
Also, first-feeding carp fed either on a phospholipids deficient diet exhibited a higher incidence of spinal deformities than larvae fed a phospholipids enriched diet. Larval ayu may require phospholipids in their diet to prevent the incidence of malformations, especially scoliosis and jaw deformities.
Postlarval P. japonicus shrimp fed a PC-supplemented diet exhibited a higher resistance to a salinity stress than animals fed a phospholipids-deficient diet. This PC effect on stress sensitivity could not be confirmed for P. vannamei, suggesting that this criterion may not be suitable for evaluating the nutritional status of euryhaline species.
The phospholipids requirement generally decreases with age or developmental stage, as shown for rock bream, where required levels decreased from 5 percent to 3 percent of soya lecithin from larva to the juvenile stage. Increasing the dietary phospholipids level beyond the required level did not affect survival or growth, as found in various studies.
However, survival of larval P. japonicus decreased when more than 3 percent soya lecithin was added to the diet. In P. vannamei lower growth rate of postlarva was observed when fed 3 percent pure PC compared to the shrimp fed 1.5 percent PC.
Several authors have suggested that larval stages are not capable of synthesizing phospholipids at a rate sufficient to meet the requirements for the formation of new cell components during the initially short period of rapid larval growth. Dietary Phospholipids enhance the transport of lipids in crustaceans, i.e., the export of absorbed lipids from the gut epithelium to the hemolymph, and the mobility of lipids between the various tissues and organs. The lack of dietary lecithin in casein diets for juvenile lobster was associated with lower hemolymph levels of phospholipids and cholesterol.
Incorporation of dietary phospholipids improves the mobilization of cholesterol and triglycerides from the gut to the hepatopancreas, hemolymph and muscle in P. japonicus. The inclusion of phospholipids in the diet affected lipid deposition, resulting in increased lipid retention in the animal. Recently it was shown that phospholipids were the more efficient mode of supply for dietary EPA and DHA to sea bass larvae.
Incorporation of 3 percent soya lecithin in the diet of P. japonicus shrimp found increased body levels of phospholipids, in particular PC, and cholesterol. Phospholipids provide choline, phosphorus and essential fatty acid for fish growth. Dietary Phospholipids has beneficial effect on growth performance indirectly through increased levels of CCK-mediated stimulating pancreatic secretion by increased level of chylomicron.
Higher dietary phospholipids (57.2–85.1 g/kg) could be beneficial for survival and growth performance of large yellow croaker (Larmichthys crocea) larvae. Higher dietary phospholipids promote development of the digestive tract and stress tolerance of yellow croaker (Larmichthys crocea) fish larvae.
Perspectives
Phospholipids are the major constituents of cell membranes and have important cellular and physio-metabolic roles, and are critical for normal functioning of cells and organs. Because larval fish and shellfish cannot synthesize phospholipids at the rates needed to satisfy their nutritional requirements, hence, phospholipid inclusion in sufficient amount in diets can ensure proper growth (including prevention of skeletal deformities), survival and stress resistance of larval fish and shellfish.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-

Amit Ranjan
Ph.D. Scholar
Department of Fish Nutrition & Feed Technology
Central Institute of Fisheries Education
Panch Marg, Off Yari Road, Versova, Andheri (w)
Mumbai - 400061, India[109,111,99,46,108,105,97,109,103,64,105,114,99,102,110,97,106,110,97,114,116,105,109,97]
Tagged With
Related Posts

Aquafeeds
The importance of carotenoids in aquafeeds
Carotenoids are important pigments that contribute characteristic quality criteria for the marketing of aquaculture products. Aquatic animals cannot biosynthesize carotenoids de novo, hence their inclusion in aquafeeds is important because they are associated with various metabolic functions.

Intelligence
Land-based macroalgae farming
Land-based cultivation of macroalgae can minimize impacts on wild macroalgae stocks while reducing harvest costs and controlling quality. Integration of macroalgae with land-based fish culture systems could reduce capital and operating costs.

Aquafeeds
New bio-processed protein concentrate shows potential
The performance of a new fishmeal replacement product was assessed for Pacific whiteleg shrimp juveniles using various experimental diets. Results indicate that this plant-based, fishmeal replacement product could be used to replace significant levels of fishmeal in shrimp aquafeed diets.

Aquafeeds
Liposomes open new doors in larval fish nutrition
Liposomes, synthetic microparticles originally developed for the pharmaceutical industry, can be produced for ingestion by rotifers and artemia. A study found that yellowtail larvae fed rotifers enhanced with liposomes containing taurine had improved growth.

Aquafeeds
Is a fish oil-free cobia feed possible?
The availability of a cost-effective grow-out feed formulation is an ongoing bottleneck for the expansion of cobia production. Studies by the authors show that the development of an aquafeed with limited or no fish oil content is possible.


