Industry efforts address control, not eradication
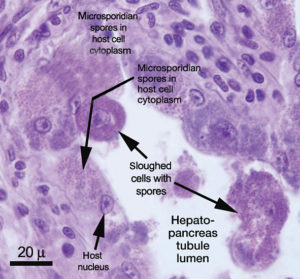
Enterocytozoon hepatopenaei (EHP), a microsporidian parasite that has been widely found in Asia and other parts of the world, is impacting aquaculture production by severely retarding the growth of cultured shrimp.
Although the most common pathology associated with microsporidians is a whitish discoloration in muscles due to spores that can stunt growth and cause other types of problems, EHP is different. It only infects the tubules of the hepatopancreas in shrimp, which damages the ability of this critical organ to gain nutrition from feed. It is widely understood that EHP does not cause mortality but heavily limits growth.
Formerly classified as protozoa, genomic taxonomy has determined that microsporidians are closely related to fungi. About 100 genera of microsporidians are known to infect crustaceans and fish.
EHP is now endemic throughout China, Malaysia, Thailand, Indonesia and Vietnam, and likely present in India and possibly Mexico. It can likely be found anywhere that has imported live feeds from China and animals from areas where EHP is endemic. EHP is very difficult to eradicate. More than likely, we will only be able to control its levels.
How is EHP detected?
The pathogen can be detected using gene-based tools such as polymerase chain reaction (PCR) and loop-mediated isothermal amplification testing of feces from broodstock. These methods can also be used with postlarvae. Light microscopy can be used, as well, although it can be very difficult to visualize the very small spores. Although effective, screening broodstock entails examination of individual animals, a costly practice. In some areas, there may be no animals totally free of the pathogen.
EHP treatment
Microsporidian infections are typically treated with a specific class of drugs that is unlikely to be effective against EHP because of its target tissue specificity. Dealing with the problem entails a three-prong strategy incorporating biosecurity in the hatchery, proper pond preparation and proper pond management during the growth cycle.
Total elimination of microsporidians may not be possible. The best approach is to lessen the loads coming into ponds and control the levels the ecosystem allows. The direct vector of EHP in ponds has not yet been identified.
Biosecurity in hatcheries
Proper practices and procedures for biosecurity in hatcheries can help control EHP.
No live feeds
Pond-reared adult broodstock, as well as those fed infected live feeds, can be infected and spread EHP through feces. The use of live animals — including polychaetes, clams, warmwater squid and locally produced Artemia – in broodstock maturation facilities poses a significant biosecurity risk and should be discouraged. Live feeds such as krill do not pose a risk. If live feeds are used, they should be frozen, pasteurized or even irradiated.
Disinfection
Maturation facilities and hatcheries should be dried out completely, washed and then disinfected with a caustic solution of sodium hydroxide. It has been suggested that all equipment, pipes and tanks should be soaked in a 2.5 percent sodium hydroxide solution for at least three hours. After this, the remaining caustic solution should be washed away, and all of the treated materials allowed to dry for an extended period.
Rinse before use with acidified chlorine at 200 ppm and pH less than 4.5. Microsporidian spores are extremely resistant to most treatments, and complete elimination will prove challenging. The goal is to substantially lower the load.
Clean eggs, nauplii
Proven strategies for washing and rinsing nauplii with the appropriate mix of freshwater and chemicals (iodine and formaldehyde, among others) that can weaken the passive attachment of spores to eggs and nauplii – thus lessening transmission – must become routine. This is an effective tool against EHP, as well as for lowering the loads of the bacteria that cause early mortality syndrome that pass from broodstock to postlarvae.
Pond preparation
High organic loads typically relate to spore loads. There is likely some intermediate vector, and until we are sure what it is, use strategies to properly treat sediments before stocking.
As spores typically are resistant to a wide variety of environmental conditions, with different species displaying differential susceptibility, the general suggestions are to physically remove accumulated organic matter and treat pond bottoms with a very caustic material to bring the pH to 12 and kill many of the spores. Killing all of them may not be possible.
It has been recommended that earthen ponds be disinfected with very heavy use of calcium oxide, or quick lime, applied at a level of 6,000 kg/ha or greater. Pond bottoms must be completely dry. Plow the quick lime into the dried sediments to a depth of 10 to 12 cm, then moisten the sediments to activate the lime. If the application is done properly, the pH of the soils will rise to 12 or more within days and then gradually return to normal as the quick lime becomes calcium carbonate.
Pond management
After the soils have recovered, use suitable commercial products from the early stages of culture to prevent the accumulation of large amounts of organic matter. These can be used alone or in combination with water exchange.
The goal is to lessen the amount of accumulated organic matter and thus reduce the potential reservoir for spores that will be ingested and continue to infect shrimp. Consistent use at levels that lessen the amount of organic matter is important.
Perspectives
Reducing the load of spores in the production environment by reducing their false vertical transmission as a result of contaminated surfaces due to spawning, in combination with aggressively limiting the reservoirs for spores, will reduce the severity of EHP. Consistent use of these methods should lessen the long-term impacts and reduce the environmental load of spores.
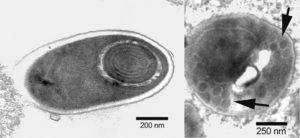
Editor’s Note: Dr. Tim Flegel kindly provided the following additional comments regarding detecting EHP, as well as the photos that illustrate this article.
Microscopy can be used to detect EHP with hepatopancreas tissue smears or tissue sections, but scanning using at least a 40x objective is needed, followed by confirmation by inspection using a 100x objective. Since this is very tedious and also depends on seeing mature or nearly mature spores, the process can take a lot of time and may fail if no spores are seen. This often happens with Litopenaeus vannamei.
Thus, PCR detection is preferred, using DNA extracted from hepatopancreas tissue as the best option. With nested PCR, DNA from feces can also be used, but may give negative results if the tested shrimp have light infections.
(Editor’s Note: This article was originally published in the March/April 2015 print edition of the Global Aquaculture Advocate.)
Author
-
Stephen G. Newman, Ph.D.
President and CEO
AquaInTech Inc.
6722 162nd Place Southwest
Lynnwood, Washington 98037-2716 USA[109,111,99,46,104,99,101,116,45,110,105,45,97,117,113,97,64,109,119,101,110,103,115]
Related Posts

Health & Welfare
Building a better shrimp nursery, part 1
Shrimp nursery systems offer an important opportunity to increase profits. Properly designed and operated nurseries are highly biosecure facilities to grow postlarvae at very high densities.

Health & Welfare
EHP a risk factor for other shrimp diseases
Laboratory challenges and a case-control study were used to determine the effects of EHP infection on two Vibrio diseases: acute hepatopancreatic necrosis disease (AHPND) and septic hepatopancreatic necrosis (SHPN).

Health & Welfare
Cost-effective biosecurity crucial for shrimp farming
Many shrimp producers give only perfunctory attention to routine biosecurity at hatcheries and farms. A cost-effective biosecurity program for farmed shrimp requires reliable diagnostic tools to make timely decisions to control or exclude pathogens.

Innovation & Investment
GOAL 2017 recap: The aquaculture opportunity has hurdles to clear
The great aquaculture opportunity – assuming responsibility as a reliable and predictable source of nutrition for a growing global population – would be better accomplished with a greater sense of confidence, according to keynote speaker Pearse Lyons.


