Results show effects of type of peptides, content of low molecular weight fractions, and resulting free amino acids

The Pacific white shrimp (Litopenaeus vannamei) is one of the fastest-growing and most profitable aquaculture species globally, with 2018 production reaching 4.9 million metric tons. The farmed production of L. vannamei represents 70 percent of the global, cultured shrimp production, and had a value of (U.S.) $35 billion in 2019. In the case of Ecuador, the country went from producing 550,000 MT in 2018 to 663,073 MT in 2020 and globally ranked as the top L. vannamei exporter last year.
Formulated aquafeed is one of the key factors in the growth of the aquaculture sector. However, one of the most complex and challenging issues for the aquafeed industry is reducing its use of ingredients of marine origin while increasing feed sustainability. Advances in feed formulation directly benefit farmed shrimp production and are critical to its profitability, as manufactured aquafeeds represent 40 to 60 percent of production costs.
With the growing interest in formulating more aquafeeds based on vegetable proteins and alternative sources to replace fishmeal, there are challenges regarding the attractive properties of these feeds, as some vegetable protein sources do not have the same attractability when compared to fishmeal. Attractability is directly related to chemoreception, an extremely well-developed and acute sensing mechanism in many marine species. However, there is limited information regarding the specific nature of the substances that elicit chemically mediated responses to food in several farmed species. Various marine protein hydrolysates have been widely used cost-efficiently and providing attractability to aquafeeds.
This article – adapted and summarized from the original publication (Molina Poveda, Cesar y Manuel Espinoza Ortega. 2021. Los hidrolizados como ingredientes funcionales en el alimento para camarón blanco. Aquacultura No. 140: 50-55) – discusses the potential for the use of hydrolysates in shrimp feeds, the hydrolysis process and its relevance for the final product quality and presents some trial results on the use of hydrolysates in shrimp feeds.
Hydrolysis
Hydrolysis is a process by which the chemical bonds between the amino acids that make up proteins are “cut.” The resulting hydrolysates are low molecular weight fractions that favor the absorption of nutrients, making them more digestible and also improving functional properties such as attractability.
Hydrolysates not only have attractant activity but are also the basis for the development of antioxidants, immunomodulators, emulsifiers, flavoring agents and antibacterial agents. For example, antioxidant activity has been reported in both fish and shrimp hydrolysates which neutralize the action of free radicals [unstable molecules or atoms that can damage the cells in live organisms]. The compounds responsible for these antioxidant properties are released after the endogenous (internal) or exogenous (external) enzymatic hydrolysis of proteins.
Regarding their antibacterial activity, Dr. George Flick reported that anchovy hydrolysate made with the enzyme pepsin had a greater antibacterial effect than other hydrolysates obtained by the action of other enzymes like papain, trypsin, alkaline protease or acid protease. The mode of action is by effectively inhibiting the growth of various bacteria – Escherichia coli, Pseudomonas fluorescens, Proteus vulgaris, and Bacillus megaterium – with minimum inhibitory concentration, MIC [lowest concentration of a chemical that prevents visible growth of a bacterium or bacteria] values of 28, 38, 56 and 75 μg/ml, respectively.
Hydrolysis is done using chemical and enzymatic methods. In the first one, alkalis or acids act on the molecular bonds and produce smaller protein fractions. Enzymes are added after heat treatment of the by-product to denature endogenous enzymes (Fig. 1). Although chemical hydrolysis is comparatively cheaper and simpler than enzymatic hydrolysis, it has several disadvantages, including its difficult control and the generation of highly variable hydrolysis products in terms of quality.
In enzymatic hydrolysis (Fig. 2), the raw material is first thermally treated and then exogenous enzymes are added from a wide variety of sources ̶ animal, plant or microbial. Enzymatic processes have become more prevalent for protein hydrolysis because of their versatility and the possibility of establishing very precisely the “size” of peptides [short chains of between two and fifty linked amino acids] required without impacting the nutritional quality of the product but rather improving it. Hydrolysis with exogenous enzymes can therefore be comparatively faster and more controllable than chemical hydrolysis.
Protein hydrolysis processes result in amino acids and low molecular weight fractions, with the enzymes involved “cutting” molecular bonds at specific sites. The enzymatic processes are especially influenced by the pH, time duration and temperature of the reaction, which determine the degree of hydrolysis and the quality of the products obtained.
https://www.aquaculturealliance.org/advocate/protein-hydrolysates-larval-fish-nutrition/
Hydrolysates from byproducts of terrestrial animal origin
One of the most widespread strategies to manage processing residues and waste from the terrestrial meat food industry is the use of the byproducts generated as animal feed ingredients by transforming them into compounds of high nutritional value. Once these residues have been dehydrated and ground, they can be used through different processes such as hydrolysis.
In 2020, Soares, et al. reported digestibility and attractability tests in diets for juveniles of L. vannamei with a protein hydrolysate from avian residues (CPH) and with a combination of this hydrolysate plus swine liver (PHPPL). These two raw materials were used as a substitute for salmon by-products. The study results revealed that the two hydrolysates had higher digestibility for protein, dry matter, energy and essential amino acids than the control sample, while no differences were found in the attractability test. In a second phase of this same study, the PHPPL combination was used in a growth trial for 42 days, with replacements of 0, 25, 50, 75 and 100 percent. No differences in shrimp survival were found between the different treatments; however, better shrimp growth was shown for the diet with 25 replacement compared to the control.
Hydrolysates from byproducts of marine origin
Waste generated by the fish processing industry can be a significant problem, but properly processed can also become valuable ingredients that contribute to the economy and sustainability of the industry. In the manufacturing of shrimp aquafeeds, certain marine ingredients (squid, tuna, crustaceans), when hydrolyzed under the correct conditions, can generate a high level of nitrogenous components that are highly palatable and contain bioactive, nutritional and functional properties that improve farmed shrimp performance.
Crustacean hydrolysates come from body parts like the head and exoskeleton (shell) often considered as waste, so their use constitutes a practical way to avoid waste and convert these body parts into valuable ingredients which, depending on the transformation process used, can produce molecules with antimicrobial, antioxidant and attractant properties. Several researchers (information available in the original publication) have reported on the use of marine hydrolysates and their positive effects on shrimp growth, survival, immunity and other benefits.
Experimental trials
Case study 1
Using a recirculation system designed for L. vannamei juveniles, we evaluated the growth and feeding efficiency of eight diets prepared with equal inclusion levels of both marine hydrolysates and hydrolysates from terrestrial animals. As a control, a non-hydrolyzed ingredient (fish soluble) with attractant properties was used. The diets were formulated to meet established nutritional requirements according to the NRC (National Research Council) guidelines and manufactured on a laboratory scale. The raw materials were ground to ensure a suitable particle diameter (<250 µm) and then pelletized.
Hydrolysates A, B, C, D, and E were obtained from marine sources such as fish and shellfish waste, while hydrolysates F and G were derived from terrestrial animal by-products; all hydrolysates were of commercial quality. Regarding the hydrolysis processes used to produce these hydrolysates, they were classified according to the degree of hydrolysis used as high, medium and low degree of hydrolysis (Table 1).
Boyd, FCR, Tabla 1
| FCR | N (kg/t) | P (kg/t) | Demanda de oxígeno (kg/t) |
|---|---|---|---|
| 1.0 | 24.7 | 8.0 | 1,100 |
| 1.1 | 30.0 | 9.1 | 1,210 |
| 1.2 | 35.4 | 10.2 | 1,320 |
| 1.3 | 40.7 | 11.4 | 1,430 |
| 1.4 | 46.0 | 12.5 | 1,540 |
| 1.5 | 51.4 | 13.6 | 1,650 |
| 1.6 | 56.7 | 14.7 | 1,760 |
| 1.7 | 62.0 | 15.8 | 1,870 |
| 1.8 | 67.3 | 17.0 | 1,980 |
| 1.9 | 72.7 | 18.1 | 2,090 |
| 2.0 | 78.0 | 19.2 | 2,200 |
Hydrolysate C was manufactured with exogenous enzymes and was subjected to a hydrolysis process that fractionated proteins to relatively low molecular weights. Hydrolysate D was produced from crustacean byproducts treated with exogenous enzymes, with a hydrolysis process considered moderate, while hydrolysate E was based on high-fat fish byproducts. Hydrolysates F and G were produced from terrestrial byproducts.
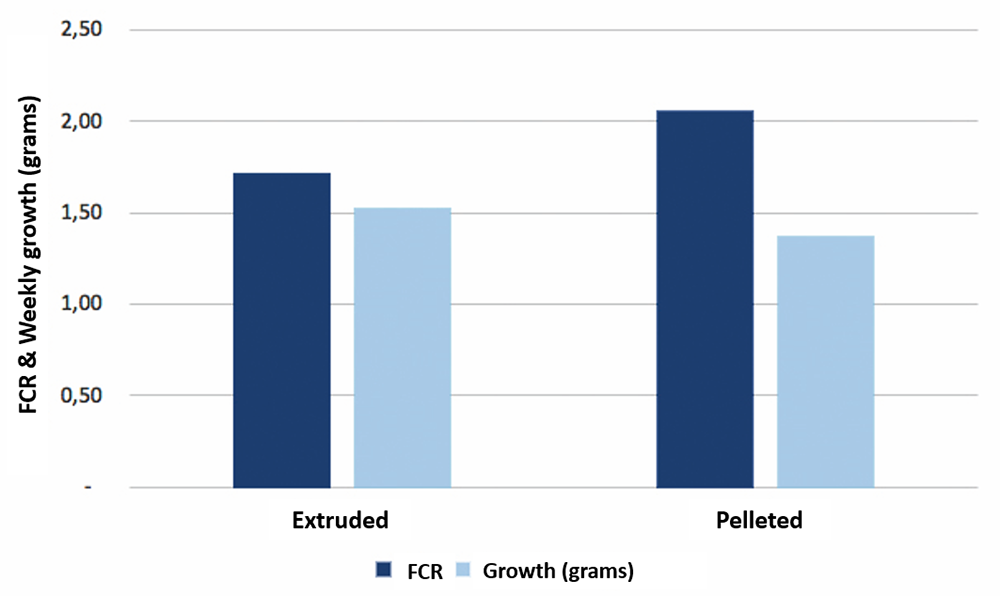
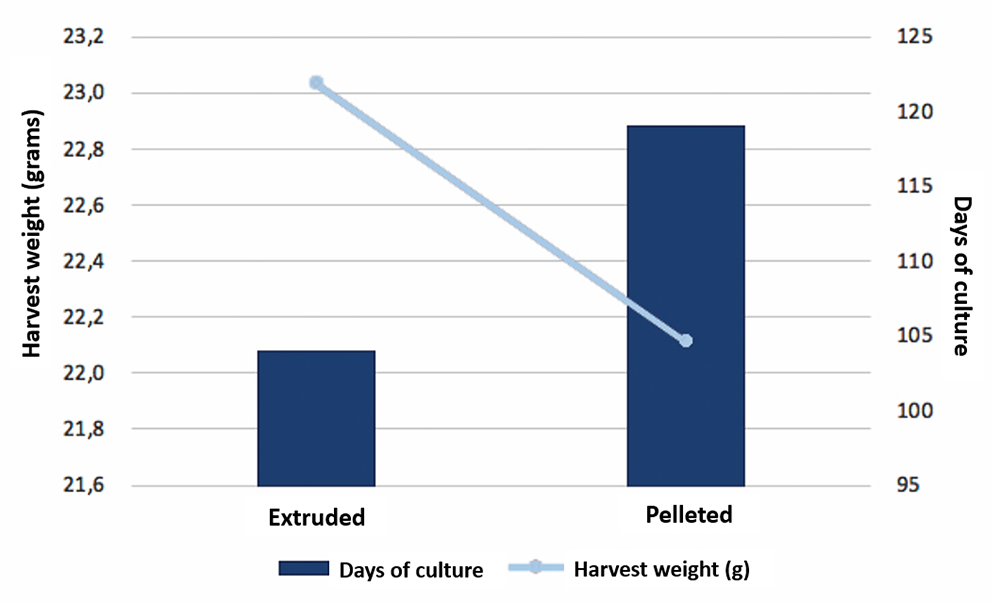
During the 61-day study, juvenile shrimp (average weight 4.36 grams) were fed ad libitum three times a day. At the end of the trial, significant differences were observed in final weight, weekly growth and FCR. Table 2 presents the zootechnical results, including shrimp survival, final weight, final biomass and weekly growth. Survival results ranged from 84 to 96 percent, with no significant differences between treatments.
CSIRO-Viet Uc, Table 1
| Parameter | Initial protocols (2018) | Final protocol (2020) | |||
|---|---|---|---|---|---|
| Experiment 1 | Experiment 2 | Experiment 6 (BFT only) | |||
| BFT | CW | BFT | CW | ||
| Yield (ton/ha/cycle) | 27.0 (24-30) | 18.5. (15-22) | 32.3 (32-33) | 24.5 (24-25) | 43.3 (39-46) |
| Survival (%) | 81.0 (70-92) | 63.5 (60-67) | 86.6 (86-87) | 66.3 (60-72) | 94.7 (82.7-99.1) |
| FCR | 1.4 (1.2-1.5) | 1.9 (1.9-1.9) | 1.3 (1.2-1.3) | 1.4 (1.3-1.4) | 1.3 (1.2-1.5) |
| TAN (mg/L)1 | 0.2 (0.9) | 2.3 (7.3) | 0.11 (0.5) | 0.54 (2.41) | 0.13 (0.30) |
| Nitrite (mg/L)1 | 12.3 (56.6) | 12.7 (44.7) | 2.66 (12.85) | 13.2 (45.3) | 1.28 (5.41) |
| Vibrio (green vibrio: total bacteria ratio)2 | 1.9 (1.3-2.6) | 7.3 (6.8-7.9) | 6.0 (5.9-6.1) | 12.0 (9.0-15.0) | 0.1 (0-0.1) |
| Water use (L/kg of shrimp) | 467 (416-519) | 6,158 (5,164-7,153) | 378 (376-380) | 4,756 (3,343-6,169) | 674 (624-812) |
Data presented as average (all ponds) and maximum and minimum (by treatment) 1 Average (all ponds) and maximum value (peak) observed in a particular treatment 2 Project target < 5%
The higher final weight for shrimp receiving the hydrolysate A treatment could be an effect of the lower survival (84 percent) observed. This hydrolysate was manufactured with endogenous enzymes, where process control is more difficult as the initial enzyme mixture is of unknown concentration. Among all the products, hydrolysate D produced the highest growth rate, biomass and survival, and we attribute this to the hydrolysate having more than 90 percent of its composition made up of protein fractions with a molecular weight less than 1000 Daltons [a Dalton, Da, is a measure of molecular weight or molecular mass; one hydrogen molecular atom has a molecular mass of 1 Da], and it also underwent a careful drying process during its manufacture. According to other researchers, the attractant fractions in hydrolysates have an average molecular weight of around 700 Da. Among the tested hydrolysates from terrestrial animals, hydrolysate F – with protein fractions with molecular weights averaging 10,000 Da – had the lowest shrimp final weight, final biomass and weekly growth.
Several studies have shown that the incorporation of hydrolysates in shrimp feeds has a positive effect on animal growth. This effect is attributed to improved absorption efficiency of the hydrolysate and to the release of amino acids and low molecular weight compounds during the hydrolysis process, which can improve the attractability to shrimp and increasing their feed intake and weight gain.
Results are closely related to the quality of the raw material and the hydrolysis process used. To obtain a functional ingredient with specific protein fractions, control of the hydrolysis process is very important – including process temperature, pH, time and others – so that the enzymes used can act as desired while prioritizing the generation of certain components that have beneficial properties for the target species.
Case study 2
We also evaluated the effect of the inclusion of different types of commercial attractants in L. vannamei juveniles (4.13 ± 0.11 grams) in a recirculation system. Five experimental diets were prepared in the lab with the inclusion of the following commercial products: pheromones (FN); a mix of natural and artificial attractants (MNA); marine hydrolysate (HM); a mix of free amino acids (AL); and fish soluble (FS) in the inclusion doses recommended by each manufacturer. The diets were designed to meet the nutritional requirements of L. vannamei, with 35 percent protein and 6 percent lipids.
The shrimp were manually fed to satiation three times a day for 55 days. At the end of the experiment, the final weight, survival, biomass, weekly growth and FCR were statistically analyzed (analysis of variance, ANOVA) to determine any significant differences (p <0.05) between the diets. Water quality parameters during the trial were monitored daily, with dissolved oxygen levels maintained above 5 ppm, temperature at 28.9 ± 0.9 degrees-C and salinity at 29.9 ± 0.5 ppt.
With the exception of survival, statistical differences were found in the zootechnical performance results. The shrimp group receiving the diets with ingredients of marine origin (FS and HM) had the best results compared to diets with the other ingredients with attractant properties (FN, MNA and AL).
Among diets with marine attractants, our results showed that the HM diet had better results compared to the FS diet in terms of final weight (10.94 vs. 10.12 grams, respectively), as shown in Table 3. The diets including FN, MNA and AL had similar zootechnical results when compared to each other. The lowest FCR was observed in shrimp fed the HM diet, which was significantly different from the FN and MNA diets.
Shinn, Bioseguridad de alimentos acuícolas, Tabla 1
| Nombre | Organización |
|---|---|
| Alexander Döring | European Feed Manufacturers Federation (FEFAC) |
| Carlos Zarza | Skretting - Norway |
| Craig Browdy | Zeigler Brothers Inc - USA |
| Hans Nauwynck | Laboratory of Virology, Ghent University - Belgium |
| Javier Ovieda | Federation of European Aquaculture Producers (FEAP) |
| Jie Huang | Network of Aquaculture Centres in Asia-Pacific (NACA) |
| Kallaya Sritunyalucksana | National Science and Technology Development Agency (NSTDA) - Thailand |
| Luis Conceição | SPAROS Lda - Portugal |
| Ole Christensen | BioMar A/S - Denmark |
| Olivier Decamp | Grobest Group - Taiwan |
| Patrick Sorgeloos | Laboratory of Aquaculture and Artemia Reference Center, Ghent University – Belgium |
| Ricardo Mello | SyAqua Siam Co. Ltd. - Thailand |
| Ronald Faber | Alltech-Coppens |
| Sam Valgaeren | Bern Aqua NV - Belgium |
| Sofia Morais | LUCTA SA - Spain |
| Sudhakar Govindam | Alltech - India |
| Zhang Lu | Tongwei Co Ltd - China |
Perspectives
Our study results show different responses depending on the protein hydrolysate evaluated, which could be related to the raw material used as a substrate and the process used to achieve a certain degree of hydrolysis. This shows that the functionality of the product is based on the type of peptides resulting from hydrolysis, the content of low molecular weight, and the free amino acids obtained.
The cost-benefit, availability, quality and efficiency are all factors that must be considered when choosing a hydrolysate as a functional ingredient for aquafeeds for L. vannamei. Hydrolysates are definitely a part of the solution for improved efficiency and sustainability of the shrimp aquaculture industry.
References are available from the corresponding author.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
César Molina Poveda, Ph.D.
Corresponding author
Investigación y Desarrollo – Skretting Ecuador[109,111,99,46,103,110,105,116,116,101,114,107,115,64,97,110,105,108,111,109,46,114,97,115,101,99]
-
Manuel Espinoza Ortega, M.Sc.
Investigación y Desarrollo – Skretting Ecuador


![Ad for [Aquademia]](https://www.globalseafood.org/wp-content/uploads/2025/07/aquademia_web2025_1050x125.gif)
