Oral vaccination just as effective as an injectable vaccine

The Nile tilapia (Oreochromis niloticus) is a very important fish species ranked second only to carps in terms of global fish production from aquaculture. As many other farmed species, it is likely to face pathogenic bacteria – such as Streptococcus spp., Flavobacterium, Edwardsiella spp., Francisella spp. and Aeromonas spp. – during culture, causing high mortality rates and substantial economic impacts. There is a need to find alternative strategies to combat the risk of bacterial infections in fish cultures. Vaccines can provide long-lasting protection against pathogenic bacteria and viruses and are now widely used to control infectious diseases in many aquatic organisms.
A wide range of vaccines have been developed to protect Nile tilapia against infectious diseases; these include live attenuated vaccines against Streptococcus agalactiae; polyvalent vaccines against diseases like streptococcosis or lactococcosis; various novel vaccines for streptococcosis; and vaccines based on formalin-killed bacteria. Vaccines can be administered in different ways, but the most common is by immersion, orally as a feed additive and direct injection. The method of administration depends on fish size and stage of development as well as the nature of the pathogen being vaccinated against.
Historically, vaccines have been administered via injection, but this is time-consuming and difficult to deliver to young fish. On the other hand, while oral and immersion vaccines are simple to administer with minimum stress to the fish, they often result in limited immune responses. Improving immersion vaccines would go a long way to reducing the incidence and severity of infectious diseases on fish farms.
This article – summarized from the original publication [Linh, N.V. et al. 2022. Efficacy of Different Routes of Formalin-Killed Vaccine Administration on Immunity and Disease Resistance of Nile Tilapia (Oreochromis niloticus) Challenged with Streptococcus agalactiae. Fishes 2022, 7(6), 398] – reports on a study comparing the efficacy of vaccines prepared from formalin-killed S. agalactiae that were administered via immersion, injection and oral routes on skin mucus and serum immune response, relative immune gene expression and the resistance of Nile tilapia to infection by S. agalactiae.
Study setup
Healthy Nile tilapia were purchased from a commercial hatchery in Chiang Mai, Thailand. The fish were fed commercial pellets for 60 days and then fed on the basal diet for 15 days. Prior to the experimental trial, 10 experimental fish were randomly selected and tested for the presence of S. agalactiae. After confirming that the fish were free of S. agalactiae, 320 fish (9.85 ± 0.35 grams) were distributed into 16 glass aquaria containing 100 liters of water at a stocking density of 20 fish per tank. The feeding experiment lasted for 15 days and had four replicates of each treatment arranged in a completely randomized design.
Vaccines prepared from formalin-killed S. agalactiae were administered to Nile tilapia via three different routes: immersion in a water-based vaccine, injection with an oil-based vaccine, and as a water-based oral vaccine. There were four experimental treatments: an unvaccinated control (Treatment T1); fish given the immersion vaccine (Treatment T2); fish injected with the oil-based vaccine (Treatment T3); and fish given the oral vaccine (Treatment T4). The fish in the unvaccinated control group (T1) were injected with 0.1 mL of phosphate-buffered saline without vaccine.
At each feeding time, a visual examination was conducted to confirm that all feed was eaten, and nothing remained in the tanks. Water temperature, pH and dissolved oxygen were maintained at 28 ± 0.23 degrees-C, 7.91 ± 0.31, and 5.32 ± 0.11 mg/L, respectively.
For detailed information on the experimental design, fish husbandry, vaccine preparation and experimental diets; sample collections, immunological assays and other analytical procedures; the challenge test; and statistical analyses, consult the original publication.
Tilapia vaccines: important disease prevention, biosecurity tools
Results and discussion
Our results showed that vaccination with formalin-killed S. agalactiae via immersion, injection, or orally stimulated a range of immune responses that enhanced the resistance of Nile tilapia to this pathogen and increased the survival rate in exposed populations. Whereas starch hydrogel-based oral vaccines produced a greater immune response than injection and immersion vaccination methods, our results for vaccination of Nile tilapia with antigen (molecule that triggers an immune response) from formalin-killed S. agalactiae indicated that the immune response from water-based oral vaccination was very similar to that from the injectable vaccine.
Similar benefits were investigated by various other researchers in Nile tilapia fed a live attenuated S. agalactiae vaccine; a polyvalent inactivated vaccine containing S. agalactiae, S. iniae, L. garvieae, and Enterococcus faecalis; a novel vaccine for S. agalactiae; a naturally attenuated S. agalactiae live vaccine; and inactivated S. agalactiae and S. iniae vaccines. The delivery of oral antigens has been reported to increase resistance in fish intestines and suppress immunity, and the results of our study indicated that water-based oral vaccines can be used without reducing the pro-inflammatory immune response in Nile tilapia during infections caused by S. agalactiae.
In addition, the destruction of antigens by stomach acid and proteolytic enzymes in the digestive system is one of the most significant barriers to oral vaccines, which needs to be tackled to improve vaccine efficacy. Several techniques are being used to protect antigens from the intestinal atmosphere for their immunogenic effects in an effort to address this challenge.
Among them, various encapsulation techniques by poly-biodegradable nanomaterials such as chitosan and others have demonstrated potential. The antigen is encapsulated by nanomaterials that can sustain the right epitope (the part of an antigen that is recognized by the immune system and antibodies) until it reaches the immunological site and is released in the intestine, considerably improving the immune response. Vaccines, on the other hand, switch on the immune system to aid in disease resistance, and the use of these strategies to control infectious diseases is gaining relevance.
Perfecting the use of adjuvants (a substance or a combination of substances used to increase the efficacy or potency of certain drugs), delivery methods and innovative technologies is needed to fulfill the demand of vaccines and ensure the safe supply of healthy fish products. It is vital for the future of the fish farming industry that vaccines can be an efficient tool for lowering the use of antibiotics in animals, therefore assisting the fight against antimicrobial resistance.
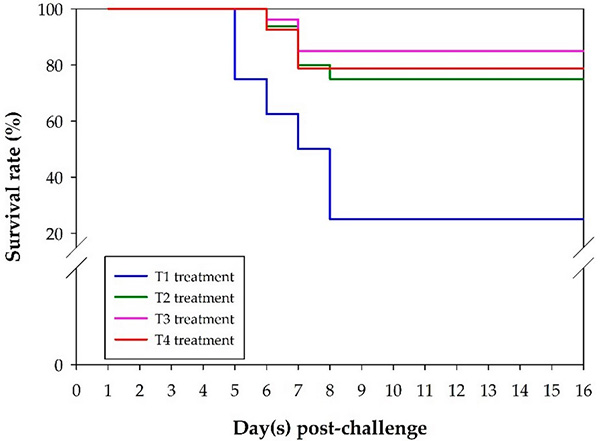
Regarding fish survival after the S. agalactiae challenge, for the unvaccinated fish in the T1 treatment group, mortality occurred on day 4 and continued until day 8, whereas the earliest incidence of fish mortality was recorded on day 5 for the vaccinated fish in the T2, T3, and T4 treatment groups. After 15 days of challenge with S. agalactiae, the relative percent survival (RPS) was 66.67 percent for the vaccinated fish in the T2 treatment group followed by 80 percent for the vaccinated fish in the T3 treatment group and 71.67 percent for the vaccinated fish in the T4 treatment group (Fig. 1). The fish that perished during the challenge experiment displayed clinical symptoms typical of S. agalactiae infection.
Vaccines have been successfully applied in fish aquaculture to protect against bacterial infections. Injection is the traditional method of administering these vaccinations, and research has shown that it is more efficient than oral administration. However, injectable vaccines in aquaculture necessitate skilled vaccinator personnel, which adds to the cost of production. Moreover, injection induces handling stress, resulting in a low feed intake and loss of appetite in fish. Following the injection of a vaccine, many fish are killed and the remaining fish may lose some of their immunity and become more susceptible to diseases. Therefore, for many fish, injectable vaccinations are not advised. Nile tilapia is one of the species that holds the most promise for vaccine development.
From our perspective, although the formalin-killed S. agalactiae water-based oral vaccine showed potential in our study to improve the survival rate and activate the immune system of experimental fish, oral nano-encapsulated vaccines are promising and practical implications for aquaculture that can offer more benefits in terms of protective efficacy, time, labor, simplicity and cost efficiency.
Perspectives
Based on our results, we suggest that the oral vaccination of Nile tilapia with formalin-killed S. agalactiae stimulated the serum and skin mucus immunity and is just as effective as an injectable vaccine. By using this kind of vaccine, farmers can save time, cut down on the demand for highly skilled labor and eliminate the stress and attendant mortality associated with direct injection. However, we recommend a larger sample size, more replicates and an analysis of a wider range of target genes as the focus of further studies.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Dr. Hien Van Doan
Corresponding author
Department of Animal and Aquatic Sciences, Faculty of Agriculture, Chiang Mai University, Chiang Mai 50200, Thailand[104,116,46,99,97,46,117,109,99,64,100,46,110,101,105,104]
Tagged With
Related Posts
Health & Welfare
Alphavirus replicon particles potential method for WSSV vaccination of white shrimp
A study demonstrated that VP19 and VP28 white spot syndrome virus envelope proteins expressed by replicon particles provided protection against mortality due to WSSV in shrimp.

Health & Welfare
Artemia bioencapsulation delivers probiotics via digestive tracts of fish larvae
Research with sturgeon and carp species indicated that encapsulated artemia has high potential to carry probiotics or other beneficial microorganisms.

Innovation & Investment
Proteon Pharmaceuticals poised to grow its bacteriophage products for aquaculture
Bacteriophage producer nets €21 million to accelerate the commercialization of its products, which aim to reduce antibiotic usage in aquaculture.

Health & Welfare
Biosecurity in intensive aquaculture: Don’t build without it
Construction of an intensive aquaculture production facility without serious attention to biosecurity nearly guarantees outbreaks of infectious disease.


![Ad for [Aquademia]](https://www.globalseafood.org/wp-content/uploads/2025/07/aquademia_web2025_1050x125.gif)
