Adding dietary soybean lecithin to marine oils could reduce the quantitative requirements of LC-PUFAs

In the past 10 years, annual world production of fish oil has remained relatively stable, ranging from 0.9 to 1.4 mmt. In contrast, aquaculture production has increased by 340 percent in the last 20 years. The demand for fish oil has continued to rise, and now 82 percent of the global supply is used in aquaculture feeds. Thus, for sustainable growth of the shrimp-farming industry, it is considered essential to reduce levels of dietary fish oil.
Concern regarding the sustainability of fish oil sources lies in the understanding that some fatty acids and other lipid classes like phospholipids, sterols and carotenoids are essential dietary components for crustaceans. On the other hand, shrimp ponds typically contain assemblages of microalgae, the nutritional value of which depends primarily on biochemical composition and the specific nutritional requirements of the animals consuming them.
Growth trial
To acquire better understanding of the contributions of nutrients from sustainable feedstuffs to shrimp nutrition in the absence or presence of natural productivity, a study was conducted in an enclosed experimental station at a shrimp farm in Tumbes, Peru. The experiment was conducted in two greenhouse-enclosed systems, one containing a clearwater system (CWS) and the other a greenwater system (GWS).
The CWS consisted of 30 indoor rectangular fiberglass aquariums with 40-L volume and 0.1-m2 bottom surface area connected to a 2,680-L semi-closed recirculating aquaculture system. The GWS containing

natural productivity consisted of 24 lined 1,944-L wooden tanks with 1.72 m2 of bottom surface and 1.13-m depth connected to an open system of three, 28-mt lined wooden reservoir tanks. The water supply for both culture systems was also used for the shrimp farm ponds.
Both systems were stocked with Litopenaeus vannamei at 80 shrimp/m2. Eight shrimp were stocked in each aquarium, while 138 shrimp where stocked in each tank.
Six experimental isonitrogenous 35 percent-protein diets were formulated with technical-grade feedstuffs to contain 10, 20 or 30 g/kg of crude Peruvian fish oil combined with 10 or 40 g/kg of 65 percent-purity soybean lecithin. Crude fat and energy levels were allowed to vary among the diets. Fast-sinking and water-stable pellets were manufactured at a pilot feed mill plant to simulate commercial conditions.
The feeding trial was conducted over 56 days by feeding shrimp in five replicate aquariums per dietary treatment in the CWS. The same six experimental diets were simultaneously evaluated in the GWS in four replicate tanks. Automatic feeders were used to supply feed five times daily with equal rations at each feeding. The feed fell only in a specially designed feed tray, which allowed daily removal and weighing of uneaten feed.
Only in the CWS were feces and molts removed daily, while bottom tank accumulation of sludge in the GWS was completely flushed on a daily basis. The feeding rates were above satiation and adjusted on a weekly basis. After termination of the feeding trial, the cephalothoraxes (fore and midgut glands) and abdominal muscles (with no intestines) of 50 shrimp/tank or 8 shrimp/aquarium were separated for analysis to compare the shrimp tissue variations among dietary treatments.
Results
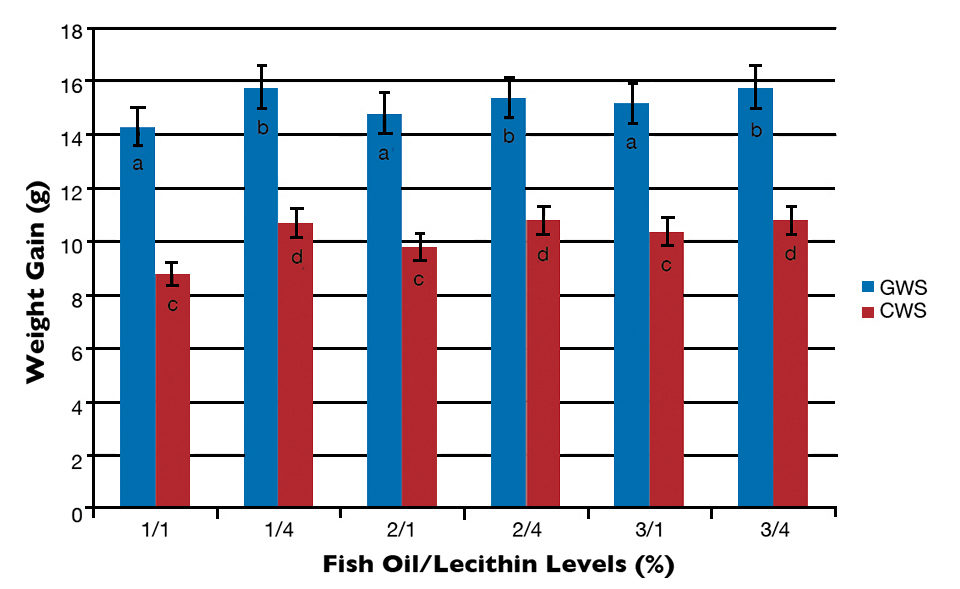
At the end of the trial, significant effects of soy lecithin level on weight gain and feed-conversion ratios (FCRs) were observed in both culture systems. However, the interaction between the fish oil and lecithin factors was not statistically significant. Shrimp fed diets containing 1 percent lecithin had significantly lower weight gain and higher FCRs than those in the rest of the treatments.
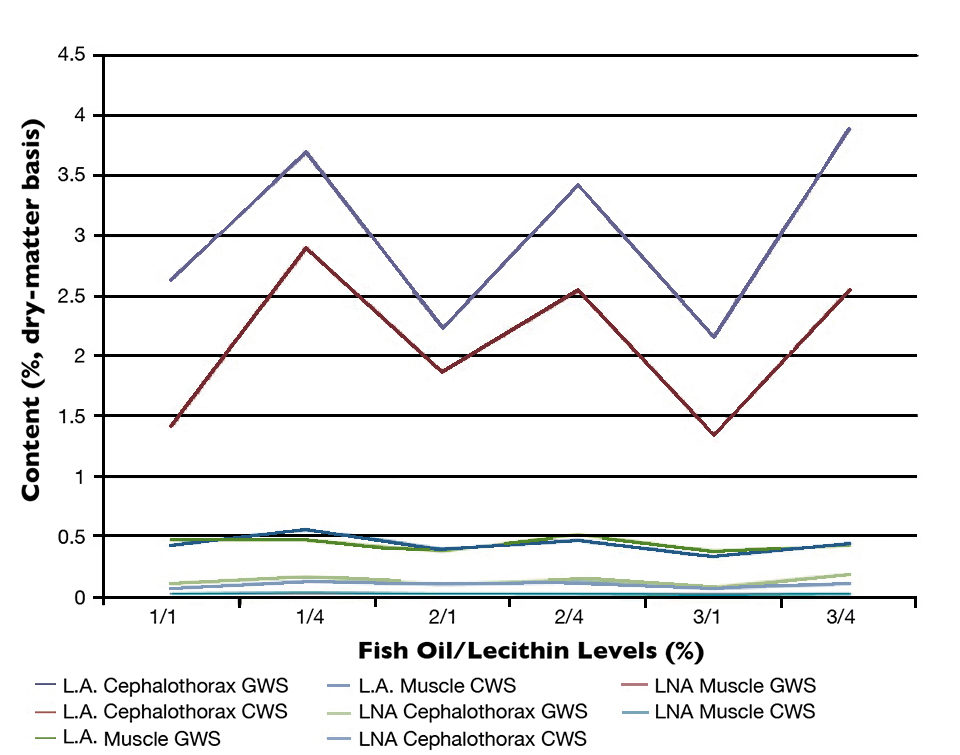
For all the dietary treatments, shrimp cultured in the GWS showed significantly higher mean lipid, linoleic acid (L.A.) and phospholipid levels in their cephalothorax tissues compared to those in the CWS (Figures 1 and 2). However, in the muscle tissues, the levels of lipid, phospholipid and linolenic acid were not different for all dietary treatments. The presence of microalgae could explain the higher levels of lipids, phospholipid, L.A. and LNA in the cephalothorax tissues of shrimp cultured in the GWS compared to the ones cultured in the CWS.
Higher dietary lecithin significantly affected the lipid cephalothorax content in both CWS and GWS (Figure 1). However, total lipid content in shrimp muscle tissue was not affected by dietary fish oil or soy lecithin levels.
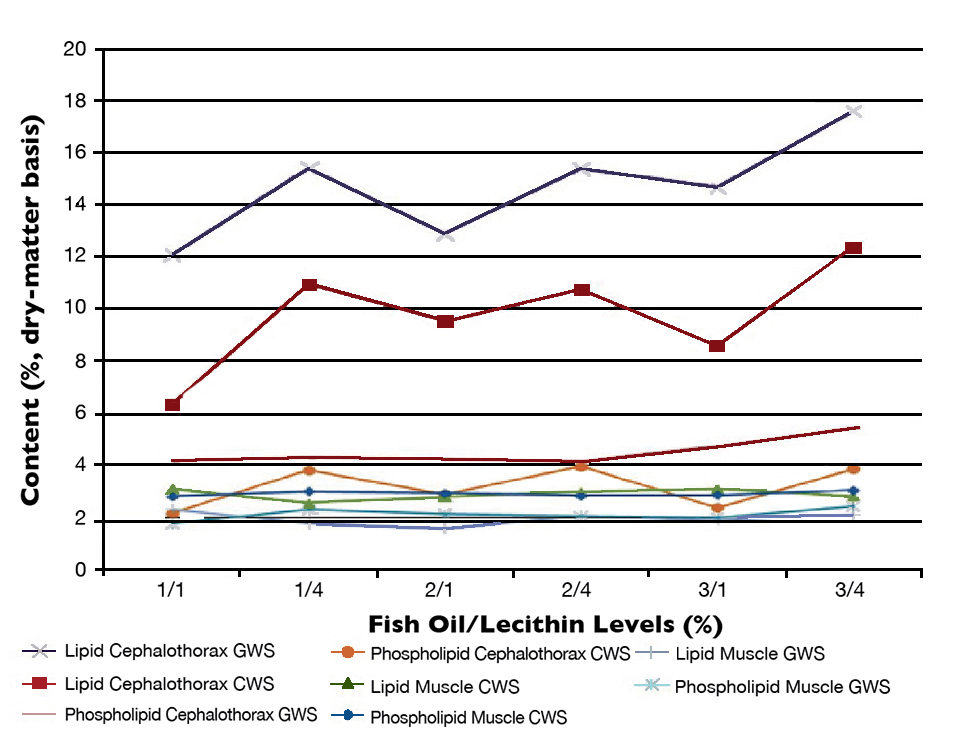
The fatty acid composition of the test diets was reflected to a certain extent in the fatty acid composition of the cephalothorax tissues of the shrimp. For instance, significantly higher levels of L.A. and were observed in shrimp cultured in both systems and fed diets containing 4 percent lecithin (Figure 2). However, no significant differences were observed in the fatty acid composition of shrimp muscle tissue.
Results from the study showed that dietary fish oil levels could be reduced to 1 percent with the inclusion of 4 percent dietary soy lecithin, suggesting that dietary phospholipid may improve the efficiency of essential fatty acid utilization when supplied as neutral lipid. Beneficial effects could be related to increased transport and lipid mobilization from the hepatopancreas to the hemolymph and other tissues and organs, resulting in enhanced lipid deposition and increased energy availability for growth.
The fact that shrimp fed diets with higher levels of phospholipid and lower levels of long-chain polyunsaturated fatty acids (LC-PUFAs) performed as well as those fed diets with higher fatty acid levels was probably because liquid soybean lecithin may serve as a source of choline, inositol, essential fatty acids or even energy.
Perspectives
The data obtained in this study indicated that shrimp weight gain in the GWS was greater than for animals of similar initial size, stocked at the same density and fed with the same diet under CWS conditions (Fig. 3). Although direct comparison between experiments is not possible because of the different system conditions, it is believed that the higher growth and performance of animals reared in the GWS could be due to their culture environment and ability to obtain continuous additional nutrients from natural food organisms present within the water column and/or pond ecosystem.
The study demonstrated that adding 4 percent dietary soybean lecithin in addition to other marine oils from squid and fishmeal could reduce the quantitative requirements of LC-PUFAs from fish oil in shrimp raised in the presence or absence of microalgae.
(Editor’s Note: This article was originally published in the September/October 2013 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Dagoberto R. Sanchez, Ph.D.
Applied Aqua Nutrition
Los Datileros MzN Lt. 10, Int. D, La Molina
Lima, Peru[109,111,99,46,108,105,97,109,103,64,48,49,48,50,99,122,101,104,99,110,97,115,100]
-
Joe M. Fox, Ph.D.
Department of Life Sciences
Texas A&M University
Corpus Christi, Texas, USA -
Delbert Gatlin III, Ph.D.
Department of Wildlife and Fisheries Sciences
Texas A&M University
College Station, Texas, USA -
Addison L. Lawrence, Ph.D.
Texas AgriLife Research
Mariculture Laboratory
Texas A & M System
Port Aransas, Texas, USA
Tagged With
Related Posts

Aquafeeds
Biofloc and clear-water RAS systems: a comparison
This study compared two types of indoor, shrimp culture systems: clear-water RAS and biofloc systems. Clearwater RAS had the edge in water quality, but shrimp in the biofloc treatment had a higher feed conversion ratio.

Aquafeeds
A new nutrient for aquaculture, from microbes that consume carbon waste
Biotechnology firm NovoNutrients aims to produce a line of nutraceutical aquafeed additives as well as a bulk feed ingredient that can supplement fishmeal. Its process includes feeding carbon dioxide from industrial gas to a “microbial consortium” starring hydrogen-oxidizing bacteria.

Intelligence
Bahamas venture focuses on grouper, other high-value marine fish
A new venture under development in the Bahamas will capitalize on Tropic Seafood’s established logistics and infrastructure to diversify its operations from processing and selling wild fisheries products to include the culture of grouper and other marine fish.

Aquafeeds
A look at phospholipids in aquafeeds
Phospholipids are the major constituents of cell membranes and are vital to the normal function of every cell and organ. The inclusion of phospholipids in aquafeeds ensures increased growth, better survival and stress resistance, and prevention of skeletal deformities of larval and juvenile stages of fish and shellfish species.


![Ad for [membership]](https://www.globalseafood.org/wp-content/uploads/2025/07/membership_web2025_1050x125.gif)
