Pacific white shrimp exposed to green light showed significantly better results in zootechnical parameters and water quality

The Biofloc Technology System (BFT) is designed to increase aquaculture productivity while improving environmental control over shrimp or fish production, reducing or eliminating water exchange, reducing effluents, minimizing farming areas size, restricting the spread of diseases, and consequently increase biosecurity. The bioflocs which make up this system consist of microbial aggregates colonized by a bacterial community, microalgae, protozoa, zooplankton, nematodes, rotifers, as well as feces and food remains.
Several factors directly influence the formation and maintenance of bioflocs, such as the carbon/nitrogen ratio, oxygenation, carbon sources, light and others. Light is considered an extremely important abiotic factor for organisms that live in the aquatic environment. Various studies have shown significant differences in the behavior, growth, food intake, maturation, reproduction and possibly changes in swimming activity of penaeid shrimp when exposed to different light conditions. Regarding microorganism populations in a BFT systems, especially when exposed to sunlight, they can often change abruptly from a heterotrophic system (dominated by bacteria and protozoa mainly) to a predominantly photoautotrophic system (dominated by microalgae).
The effect of the presence of light on shrimp farming has already been described by several authors, but it is still necessary to evaluate the effect of light separated into different wavelengths, the colors. In indoor spaces, such as sheds and buildings with farming tanks where there is no natural light and consequently artificial lighting is used, more effective control of light-related variables is possible, such as light coloration. In this article we report some recent work to evaluate the growth performance of Pacific white shrimp (Litopenaeus vannamei) experimentally reared in a BFT system with supplementary colored LED light.
The authors are grateful for the financial support provided by the National Council for Scientific and Technological Development (CNPq), Research Support Foundation of the Rio Grande do Sul State (FAPERGS) and the Coordination for the Improvement of Higher Level Personnel (CAPES). Special thanks for our sponsors, All-aqua, GUABI Animal Health and Nutrition S.A., AQUATEC and TREVISAN Aerators.
Methodology
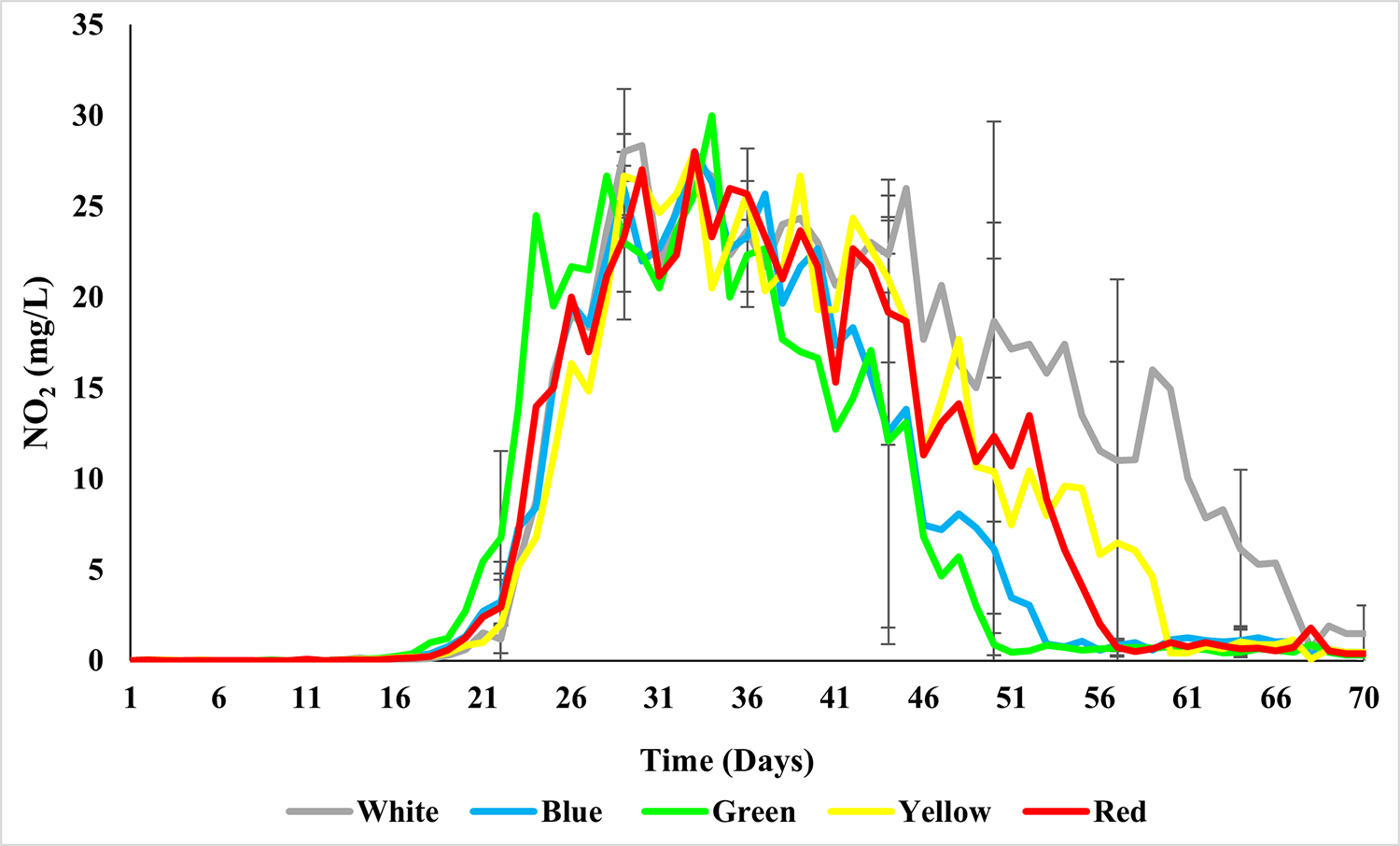
Five treatments were designed with 3 repetitions each, using LED lights in various colors, including yellow, blue, red, green and white (control). The experimental units holding the shrimp were isolated from each other using a conical lid over the tanks, so that the influence of a light color would not extend beyond those animals intended for that light color treatment. Shrimp weighing 0.37 grams were stocked at a density of 500 shrimp per cubic meter in experimental units of 150 liters, and the experiment lasted for 70 days.
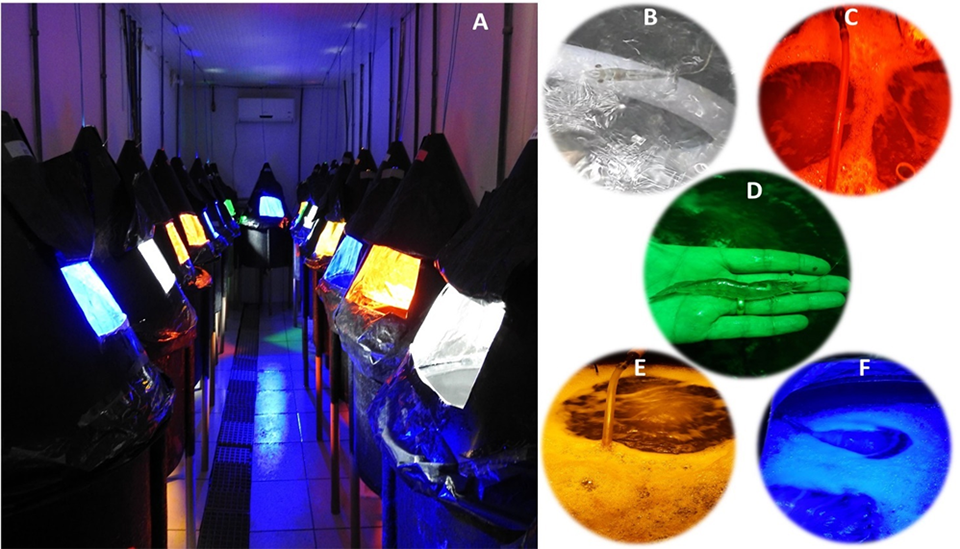
Water quality parameters such as temperature, dissolved oxygen and pH were monitored twice a day using a multiparameter probe. The concentration of total ammonia nitrogen and nitrite was quantified daily. The alkalinity was checked every three days and corrected when values below 150 mg CaCO3 per liter were observed. Water turbidity, nitrate and phosphate concentrations were determined once a week, as well as total suspended solids and settleable solids. Carbon dioxide concentrations were calculated using pH and alkalinity measurements.

Organic fertilization of the water in the experimental tanks was done using liquid molasses (C/N ratio of 6:1) whenever ammonia levels reached >1.0 mg per liter. Water exchanges were carried out when nitrite levels reached values above 26 mg per liter. Applications of a commercial probiotic (INVE® Sanolife PRO-W) to the water were performed once a week to help maintain water quality.
Shrimp were fed twice a day, using a local, commercial diet (Potimar GUABI® 38 percent crude protein). Feed adjustments were made according to the estimated growth and consumption of the feed. All results were analyzed by one-way ANOVA (α=0.05).
Results and discussion
Based on our results, significant differences were determined for water quality parameters such as nitrite, light penetration into the water column and total amount of water used during the experimental culture period. No statistical differences were found for temperature, dissolved oxygen, pH, ammonia, nitrate, phosphate, alkalinity, carbon dioxide, total suspended solids, settleable solids, turbidity and chlorophyll a.
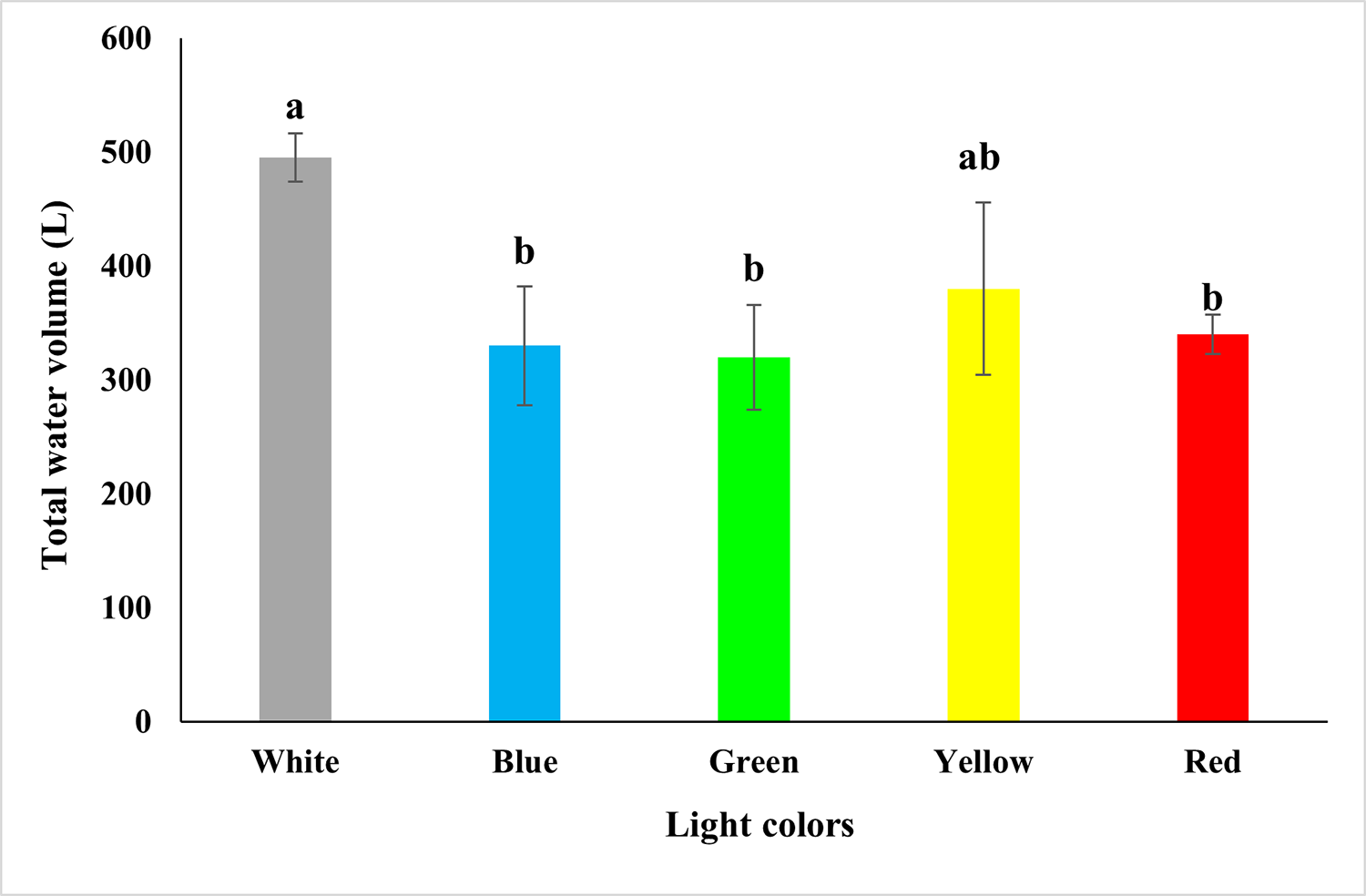
The total amount of water used to rear L. vannamei held in a BFT system with supplementary colored LED light differed significantly. The colors of green, blue and red lights used lower amounts of water, with an average of 340 liters, while the white and yellow lights had an average value of 495 liters. That is, 155 liters more to produce the same shrimp density. This difference in the total amount of water for production is related to the number of renovations through water exchanges that were carried out when nitrite levels exceeded the safety levels considered safe for the species. The green and blue light treatments had better nitrification levels when compared to the white light (control), with a difference of almost 20 days.
Gomes Reis, LEDs, Table 1
| Parameter | Green | Blue | Red | Yellow | White |
|---|---|---|---|---|---|
| Initial weight (g) | 0.37±0.20 | 0.37±0.20 | 0.37±0.20 | 0.37±0.20 | 0.37±0.20 |
| Final weight (g) | 7.59±0.30a | 6.91±0.26ab | 7.31±0.26a | 6.88±0.28ab | 6.19±0.09b |
| Survival (percent) | 92.88±1.66 | 85.77±3.82 | 90.66±2.17 | 86.22±2.73 | 90.66±2.88 |
| FCR | 1.36±0.01 | 1.62±0.14 | 1.42±0.06 | 1.58±0.02 | 1.63±0.10 |
| Productivity (Kg/m3) | 3.36±0.10 | 2.82±0.24 | 3.15±0.19 | 2.81±0.08 | 2.65±0.12 |
A significant difference in growth performance was found only in the final weight of the shrimp. The green and red colors were the only ones that were different from the white color (control). No significant differences were found for survival, apparent feed conversion ratio and productivity.
Conclusion and perspectives
The results of our study demonstrated that the aquacultured production of L. vannamei exposed to green light showed significantly better results in water quality and zootechnical parameters when compared to the results for shrimp exposed to other colors, probably due to the availability of food in these treatments and a possible photosensitivity of bacteria in different spectra of light. We are currently analyzing the microbial community and oxidative stress in the different light treatments to further elucidate various questions.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Wellica Gomes Reis
Laboratory of Microorganism in Aquaculture
Federal University of Rio Grande – FURG
Rio Grande, Brazil -
Paulo Cesar Abreu
Laboratory of Phytoplankton and Marine Microorganisms
Federal University of Rio Grande – FURG
Rio Grande, Brazil -
Hellyjúnyor Brandão
Laboratory of Carcinoculture, Marine Station of Aquaculture
Federal University of Rio Grande – FURG
Rio Grande, Brazil -
Wilson Wasielesky, Ph.D.
Laboratory of Carcinoculture, Marine Station of Aquaculture
Federal University of Rio Grande – FURG
Rio Grande, Brazil -
Dariano Krummenauer, Ph.D.
Corresponding author
Laboratory of Microorganisms in Aquaculture
Federal University of Rio Grande – FURG
Rio Grande, Brazil[109,111,99,46,108,105,97,109,103,64,107,111,110,97,105,114,97,100]
Tagged With
Related Posts

Aquafeeds
Black soldier fly larval production in a stacked production system
Study describes development and evaluation of an “all-in-one” stacked system for indoor production of black soldier fly larvae.

Intelligence
Green-lighting growth: Green LED light shows promise in flounder farming
Japanese researchers say that deploying green LED light above flounder grow-out tanks encourages rapid growth and feed intake among the fish.

Health & Welfare
How do light-dark cycles impact the physiological processes of Pacific white shrimp?
Researching the effect of light-dark cycles on various on L. vannamei physiological processes to help improve production in controlled farming systems.

Health & Welfare
Ultraviolet A photoperiod regimes have varying effects on Pacific white shrimp
Study shows that different ultraviolet A photoperiod treatments impacted growth, immunity, FCR and mortality in L. vannamei shrimp.


![Ad for [BSP]](https://www.globalseafood.org/wp-content/uploads/2025/07/BSP_B2B_2025_1050x125.jpg)
