Fishmeal can be fully replaced with BSFLM in well-balanced nursery diets for P. vannamei PLs under high-density culture

The use of insect meals in aquatic feeds has gained increased attention in recent years, driven by a greater volatility in supply and prices from conventional protein sources together with the pressing need to reduce carbon emissions and promote a circular economy. In comparison to protein sources obtained from fisheries, agriculture, and animal husbandry, the utilization of insects as feed ingredients can comprehensively integrate the advantages linked with sustainability, nutritional value, and functionality. Insects also have a short life cycle and high efficiency as food converters, requiring fewer resources for their production.
Numerous insects have been examined as potential feed ingredients for aquaculture species. Their nutrient value lies in their adequate levels of digestible protein, amino acids (AAs), lipids, and energy. A previous study with Pacific white shrimp (Penaeus vannamei) juveniles revealed that a full-fat black soldier fly (Hermetia illucens) larvae meal (BSFLM) contains crude protein (CP) and lipid content of 41.7 and 17.4 percent (dry matter basis), respectively. Other authors have reported high apparent digestibility coefficients for protein (85.1 ± 5.58 percent, mean ± SD), lipids (95.2 ± 2.43 percent) and energy (87.1 ± 6.08 percent). In shrimp feeds, the BSFLM has been the most extensively studied insect protein source. Its reported benefits have included partial replacement of fishmeal, improved intestinal health, growth and feed efficiency, higher resistance to Vibrio sp. infection and increased levels of digestive protease activity.
However, both the availability and prices of BSFLM are still a constraint for its widespread use as an ingredient in animal feeds at times where it is considered uncompetitive. But its utilization is expected to become more prevalent in specialty diets through the substitution of costly protein ingredients, such as fishmeal (FML).
This article – summarized from the original publication (Nunes, A.J.P. et al. 2023. The Black Soldier Fly (Hermetia illucens) Larvae Meal Can Cost-Effectively Replace Fish Meal in Practical Nursery Diets for Post-Larval Penaeus vannamei under High-Density Culture. Fishes 2023, 8(12), 605) – reports on a study that investigated the growth performance of post-larval P. vannamei fed nursery diets with a progressive replacement of FML for BSFLM. Additionally, a sensitivity analysis over the price of BSFLM was conducted to determine the optimal level of fishmeal replacement.
Study setup

The present study investigated the growth and economic performance of post-larval P. vannamei fed nursery diets containing graded levels of a partially defatted black soldier fly larvae meal (BSFLM). We used a progressive replacement of fishmeal for BSFLM at 0, 25, 50, 75 and 100 percent. The research consisted of a 42-day nursery culture conducted at LABOMAR (Instituto de Ciências do Mar, Universidade Federal do Ceará, Fortaleza, Brazil) to evaluate shrimp growth performance throughout post-larval and early juvenile stages. P. vannamei were purchased as post-larvae from a commercial hatchery (Aquatec Aquacultura Ltda., Canguaretama, Brazil).
For the experimental phase, shrimp were raised from PL15 to over 600 mg body weight (BW) in 50 outdoor tanks of 1.50 cubic meters (1.70-square-meter bottom area × 0.88 m height) at 1369 PLs per cubic meter (2053 ± 33 PLs per tank). Shrimp were fed five nursery diets with graded levels of BSFLM as a replacement for fishmeal at 0, 25, 50, 75 and 100 percent. One diet without BSFLM containing 22 percent fishmeal (as-is basis) served as the control. This initial set-up allowed 10 replicate tanks per feeding treatment. At harvest, shrimp survival, feed efficiency and growth performance from each tank were determined.
For detailed information on the experimental design and rearing system; animal husbandry; diet formulation and preparation; feeding and water quality; shrimp performance; price sensitivity and statistical analyses, refer to the original publication.

World’s largest fly factory attracting investors eyeing aquafeed expansion
Results and discussion
Shrimp performance in the present study was consistent with other work conducted under similar rearing conditions. For example, Nunes et al. reared post-larval P. vannamei of 3.6–2.5 mg body weight in an outdoor and indoor tank system at 2371–2504 PLs per cubic meter, respectively. After 52–41 days of nursery, shrimp reached an average final survival of 84.0–91.8 percent, 1568–1611 grams per cubic meter gained yield, 14.2–14.2 mg daily growth, and 1.70–0.89 feed conversion ratio (FCR), respectively. In their work, shrimp final BW varied according to the diet, from 683 to 775 mg and from 567 to 629 mg in the outdoor and indoor tanks, respectively.
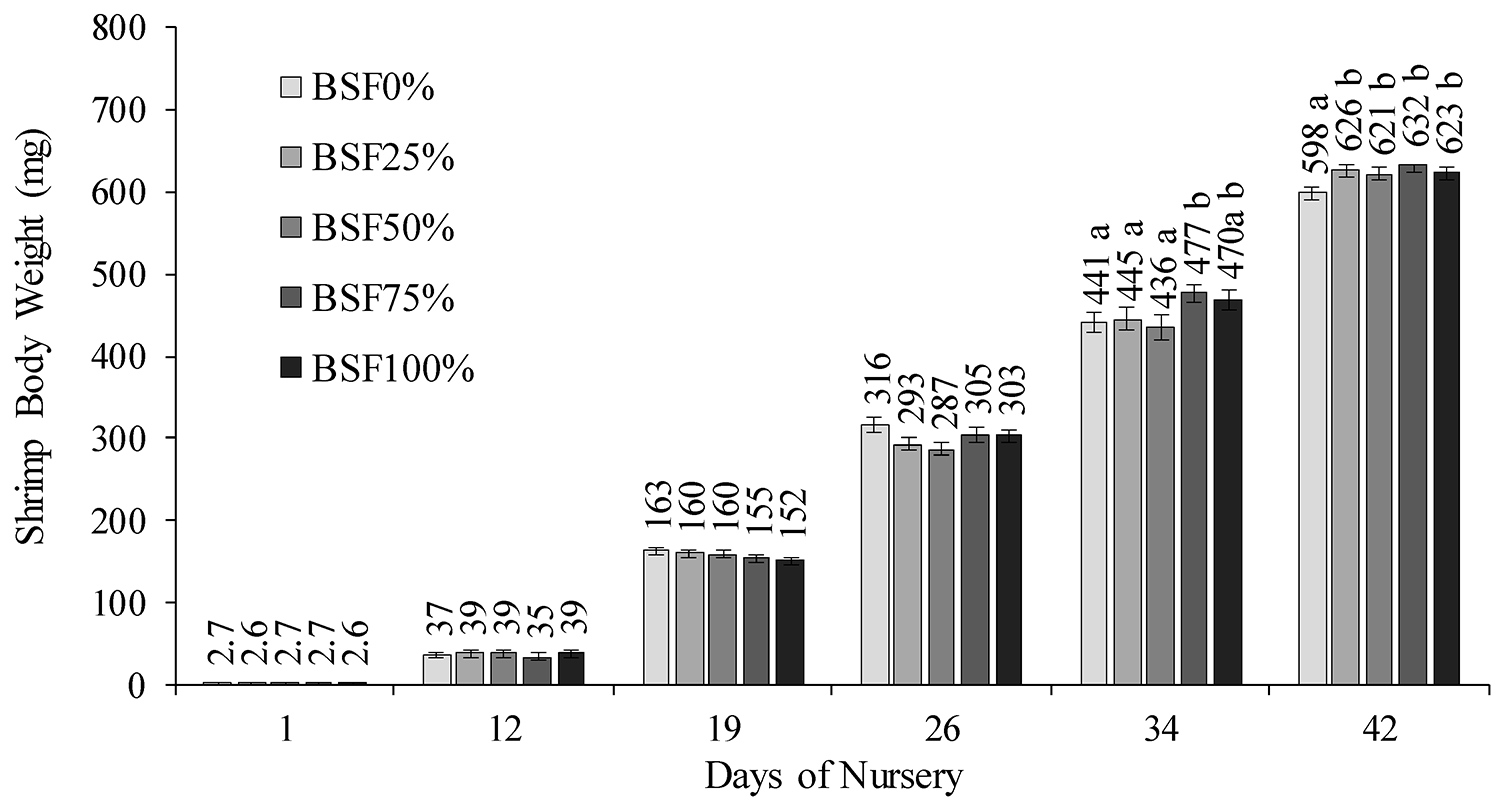
Our results here demonstrated that it is possible to fully replace fishmeal with BSFLM in nursery diets for post-larval P. vannamei with no detriment to shrimp performance. The highest dietary inclusion level obtained for BSFLM was 26.46 percent (percent of the diet, as-is) which is equivalent to a full replacement of fishmeal. This contrasts with other studies which have only reported partial replacement of fishmeal with BSFLM. For example, Chen et al. evaluated the replacement of brown fishmeal (68.21 percent CP and 9.00 percent lipid) for BSFLM at 10, 20 and 30 percent.
They reported a significant drop in final juvenile shrimp BW (from 7.76 ± 0.09 to 7.06 ± 0.16 grams) and weight gain (776.4 ± 10.3 to 698.4 ± 19.0 percent) when shrimp were fed diets with 0 and 14.25 percent BSFLM, respectively. At 30 percent fishmeal replacement, they reported intestinal cell apoptosis (programmed cell death) and degeneration. Unlike these results, our study did not identify any adverse impact on shrimp performance that could indicate potential harm to shrimp health. This could be due to the nutrient profile of the BSFLM which may vary depending on the type of residues these flies were raised on.

In our study, the favorable outcomes regarding the replacement of fishmeal were probably influenced by an appropriate supplementation with crystalline AAs, a sufficient provision of omega-3 fatty acids, and the inclusion of feed attractants in all diets. The importance of the dietary supplementation of these nutrients and feed attractants when fishmeal is challenged has been demonstrated in several other studies with penaeid shrimp.
As BSFLM inherently possesses lower levels of these nutrients compared to fishmeal, achieving full fishmeal replacement requires the formulation of a well-balanced diet. Thus, ensuring all key nutrients are appropriately supplemented in the diet is a key element to achieve full fishmeal replacement with BSFLM. Various other researchers have reported similar positive results with the replacement of fishmeal with BSFLM in diets for juvenile P. vannamei. Overall, complete replacement of FML with BSFLM is possible if diets are properly balanced for key nutrients, such as essential AAs and omega-3 fatty acids.
In our study, we observed a growth response with higher dietary inclusion levels of BSFLM, even in diets deprived of fishmeal. In terms of protein digestibility, both BSFLM and fishmeal exhibit similar characteristics. Apparent protein digestibility coefficient for BSFLM has been reported at 85.1 ± 5.58 percent (in comparison to 78.9 percent and 85.8 percent for the salmon meal, fishmeal, used in the present study). Our diets were formulated to contain nearly the same AA content, but an increase was found in the total dietary Met (Met plus cysteine) content with a progressive replacement of FML. Total dietary levels increased from 0.85 percent (1.39 percent) in the control (CTL) diet to a high of 1.03 percent (1.56 percent) in the diet with 100 percent replacement of FML. On the other hand, the CTL diet contained slightly higher levels of omega-3 fatty acids and HUFA than the diets containing BSFLM. Hence, the growth enhancement effect recorded in shrimp fed BSFLM may not have been associated with the dietary nutrient levels, which were likely above shrimp requirements.
A similar enhancement in shrimp growth performance with the dietary inclusion of BSFLM has been reported by other authors who investigated the use of BSFLM for post-larval P. vannamei (PL41, 0.1 grams initial BW). They reported that all diets with BSFLM improved weight gain, FCR and specific growth rate (SGR) compared to the diet with the highest FML level. Additionally, they reported that SGR was significantly improved from the dietary inclusion of 4.5 percent, while FCR was significantly improved at 7.5 percent. And other researchers have identified BSFLM as one of the top two options for improving the growth performance of juvenile P. vannamei (0.17 and 11.1 grams initial and final BW, respectively) among seven insect meals.
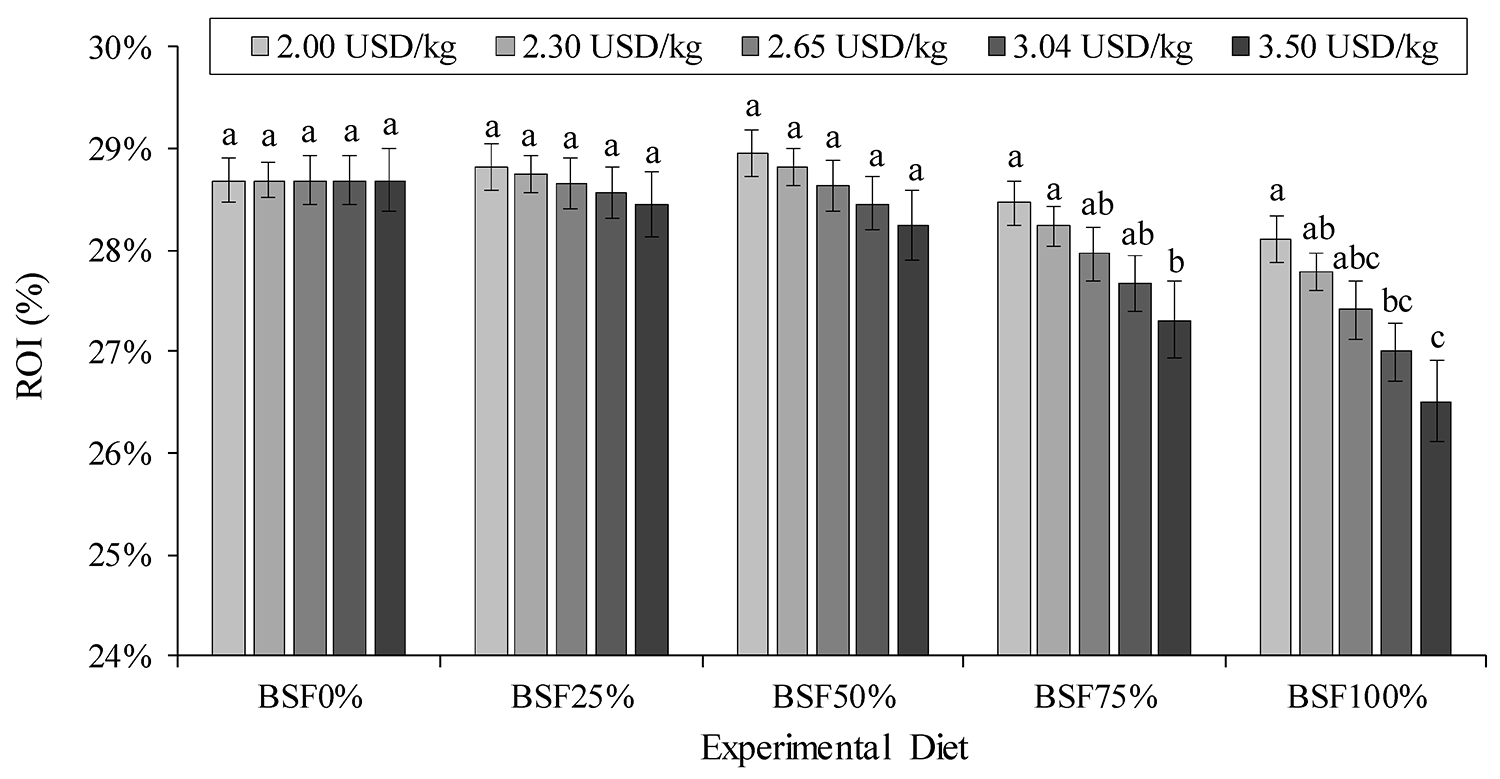
We have found that the return on investment (ROI) for farming shrimp until 23.5 grams can be impacted by the price and dietary inclusion of BSFLM in nursery diets. As the price of BSFLM rises along with its inclusion level, there is a progressive decrease in ROI. Nonetheless, a noticeable reduction in ROI occurred only when replacing more than 75 percent of FML, but this occurred when the price of BSFLM surpassed $3.04 per kg. At 100 percent fishmeal replacement, a price of $2.65 per kg for the BSFLM was still advantageous. It is important to note that these economic simulations focused solely on the impact of shrimp survival and FCR during the nursery phase in response to dietary treatment.
Shrimp BW at the end of the nursery phase also plays a crucial role in subsequent shrimp performance. For example, initiating the grow-out phase with larger shrimp can lead to shorter production cycles, early harvest, reduced operating costs, and increased annual yield. In our simulations, the operating costs associated with the use of nursery diets accounted for a small portion of the total feed costs to produce a 23.5-gram shrimp, ranging from 3.8 to 4.5 percent depending on the diet formulation. This is attributed to the minimal feed quantities used in the early stages of shrimp culture. The nursery phase allows for greater control of feed inputs due to smaller, more compact farming areas, resulting in lower FCRs compared to grow-out.
Perspectives
Based on our findings in the present study, it can be concluded that fishmeal can be fully replaced with BSFLM in well-balanced nursery diets for P. vannamei (individual weights between 2.7 and 600 mg. This corresponds to a dietary inclusion level of 26.46 percent BSFLM (percent of the diet, as-is).
While the full replacement of FML for BSFLM was successfully accomplished, it should be noted that the competitive return on investment (ROI) was sustained only when the maximum price of BSFLM did not exceed U.S. $3.04 per kg. This price level corresponds to a maximum dietary inclusion of 19.74 percent or a 75 percent replacement of fishmeal. Further research may be necessary to fine-tune cost-effective inclusion levels of BSFLM to optimize the economic outcomes while considering the fluctuating prices of fishmeal.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Alberto J.P. Nunes, Ph.D.
Corresponding author
LABOMAR—Instituto de Ciências do Mar, Universidade Federal do Ceará, Avenida da Abolição, 3207, Meireles, Fortaleza 60165-081, CE, Brazil[114,98,46,99,102,117,64,115,101,110,117,110,46,111,116,114,101,98,108,97]
Tagged With
Related Posts

Aquafeeds
Black soldier fly larval production in a stacked production system
Study describes development and evaluation of an “all-in-one” stacked system for indoor production of black soldier fly larvae.

Aquafeeds
Improving the lipid profile of black soldier fly larvae
Black soldier fly larvae fed with marine-based substrates displayed an improved lipid profile with higher polyunsaturated fatty acid levels.

Health & Welfare
Dietary evaluation of black soldier fly larvae meal on growth and health of Pacific white shrimp
Inclusion of black soldier fly larvae meal, to replace fishmeal in L. vannamei diets, improves shrimp health and disease resistance.

Aquafeeds
Meta-analysis of the effects of black soldier fly meal on fish growth
Results indicate it is feasible to achieve high levels of substitution of fishmeal with black soldier fly meal without risking a negative impact.


![Ad for [membership]](https://www.globalseafood.org/wp-content/uploads/2025/07/membership_web2025_1050x125.gif)
