Results indicate that dietary tartaric acid can promote the growth performance and immunological indicators of L. vannamei

The physiological activities of animals can be affected by intestinal microbial communities. Organic acids (OAs) are compounds with antimicrobial properties that can modulate intestinal microflora, inhibit the growth of opportunistic pathogens (e.g., Vibrio sp., Aeromonas sp.), and provide other benefits. In fact, the antibacterial mechanism of OAs acts by lowering the cytoplasmic pH of bacteria and disrupting normal cellular reactions.
Earlier reports indicated that diets containing OAs such as citric acid, formic acid, and succinic acid effectively improved immune and antioxidant responses in L. vannamei when compared with the un-supplemented group. OAs promote the disease resistance of animals by controlling the gut microbial population and stimulating the immune system.
The OA tartaric acid (TA) is a chemical compound obtained from biological (fermentation) and chemical processes that is also naturally present in different fruits such as grape, banana, lychee, sweet cherry, avocado, and tamarind. TA has gained growing popularity in food and pharmaceutical activities due to its various biological properties. Such properties include regulating acidity, improving the shelf life and sensory properties of food, stimulating the immune system, and displaying antibacterial, antioxidant and anti-inflammatory effects.
The bacterium Vibrio parahaemolyticus is a well-known opportunistic pathogen in the marine aquaculture industry and is associated with the emergence of serious acute hepatopancreatic necrosis disease (AHPND) in farmed shrimp. One of the effective strategies to increase the resistance against vibriosis infection and boost other physiological processes in farmed shrimp is the administration of OAs in their diet.
This article – summarized from the original publication (Yousefi, M. et al. 2024. Dietary Tartaric Acid Improves Growth Performance, Gut Microbiota, Digestive Enzyme Activities, Hemolymph Immunity, Antioxidant Markers, and Disease Resistance against Vibrio parahaemolyticus in Pacific White Shrimp. J. Mar. Sci. Eng. 2024, 12(1), 83 – reports on a study to evaluate the potential benefits of dietary tartaric acid on growth and nutritional markers, gut microbiota profile, digestive enzymes, immunohemocyte and antioxidant responses of Pacific white shrimp (Litopenaeus vannamei), and survival rate following an immersion challenge with V. parahaemolyticus.
Study setup
This work was conducted as a completely randomized design in a laboratory located in Bardstan (Dyer City, Bushehr Province, Iran). The effects of dietary TA on growth indices, gut microbiota, digestive enzyme levels, antioxidant and immunological markers in L. vannamei, and survival rate following a 14-day immersion challenge with V. parahaemolyticus were investigated. Six hundred shrimp (3.26 ± 0.05 grams) from a commercial supplier were stocked into 15, 300-liter fiberglass tanks at 40 animals per tank. The shrimp were fed experimental diets supplemented with five distinct concentrations of TA including 0 (TA0), 2.5 (TA2.5), 5 (TA5), 7.5 (TA7.5), and 10 g/kg (TA10) for 56 days.
At the end of the experiment, all shrimp were counted and weighed, while growth performance parameters, feed conversion ratio, protein efficiency ratio, and survival rate were calculated.
For detailed information on the experimental design, animal husbandry and diet preparation; and all analyses carried out, refer to the original publication.
Results and discussion
In recent years, the acidification of aquafeeds by the addition of OAs has been shown to provide several benefits for the digestive system health and growth of various cultured aquatic animals. In the study reported here, diets fortified with TA at 5–10 grams per kg significantly improved weight gain (WG), final weight (FW), specific growth rate (SGR) and feed conversion ratio (FCR). Comparable results were reported for L. vannamei fed diets with succinic acid at a concentration of 5 grams per kg, resulting in higher FW and SGR and lower FCR; and also for L. vannamei fed with a diet supplemented with 2 percent potassium diformate (KDF).
Several studies have reported that the dietary inclusion of OAs improved nutrient digestibility, protein and energy storage, leading to a notable increase in protein efficiency ratio (PER), FCR and SGR in shrimp. Other researchers have reported that dietary OAs increase the availability of nutrients by lowering the pH. This action is accompanied by chemical reactions that facilitate the absorption of phosphorus and other minerals.
In addition, OAs participate in metabolic activities thereby increasing the generation of adenosine triphosphate (ATP; a nucleotide that provides energy to drive and support many processes in living cells), which in turn improves growth performance and feed utilization.
Researchers have demonstrated that the acidification of aquafeeds with OA could regulate the gut microbiota by increasing beneficial strains resistant to acidity, like lactic acid bacteria (LAB) and breaking down bacteria sensitive to low pH. Previous reports have revealed that gut LAB plays a vital role in the modulation of gut enzymatic capacity, thereby enhancing the growth performance of the host.
In this study, the inclusion of TA in experimental diets at 5–10 grams per kg promoted the abundance of indigenous LAB in the intestine, which was associated with the increase in intestinal digestive enzyme activity and growth performance. Similarly, other findings indicated that enhancing the LAB population led to improving digestive enzyme activity in L. vannamei and other aquatic species.
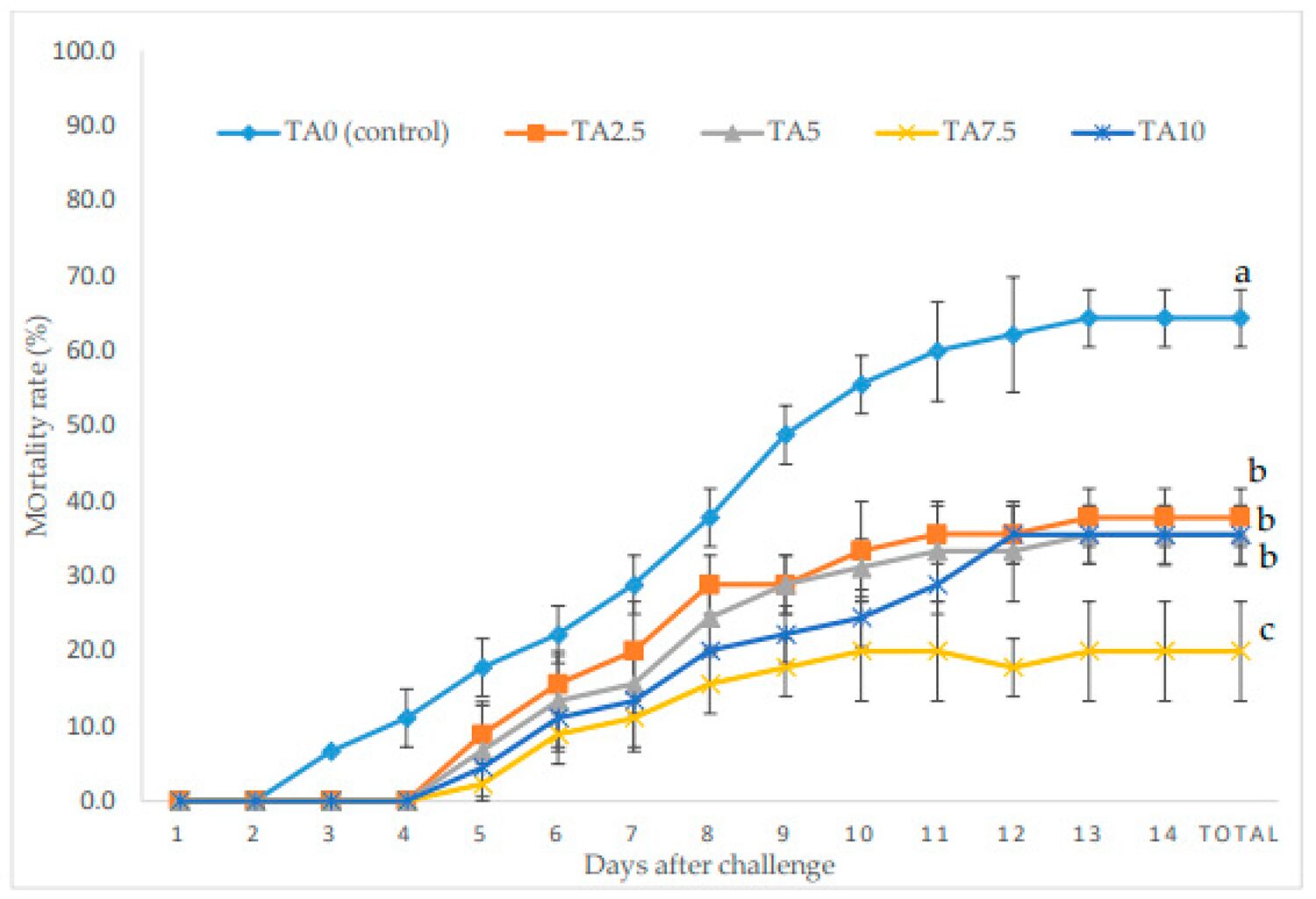
Regarding the challenge test, the mortality rate (MR) of L. vannamei exposed to V. parahaemolyticus during a 14-day challenge is shown in Fig. 1. At the end of the challenge period, all supplemented groups of shrimp had a much lower MR compared to the control group, TA0. The lowest MR was observed in the TA7.5 group, with significantly lower MR than the other TA-treated groups.
Shrimp and other crustaceans lack evolved specific immune responses and, unlike fish, rely mainly on several innate immune responses, so promoting the innate immune system of farmed shrimp is particularly important. Circulating hemocytes (cells involved in the immune system of invertebrates and found within the hemolymph) are the main mediators in creating cellular and humoral responses in crustaceans.
Determining hemocyte counts is one of the most reliable indicators to determine the effect of manipulated diets on immunity. In the present research, the total hemocyte count (THC) in shrimp markedly increased in all TA-supplemented groups, which could be related to the maximum release of minerals such as copper and iron. Consistent with our results, other researchers have reported that feeding L. vannamei with the OA sodium butyrate significantly improved the shrimp THC counts. In another study, the THC count was reported to be higher in L. vannamei fed diets supplemented with formic acid and the pigment astaxanthin.
Enhancing the enzymatic or non-enzymatic reactions of the antioxidant system during the culture period plays a vital role in improving the health of crustaceans against biological or non-biological stress factors. The inclusion of OAs in the diet is one of the most practical and effective techniques used to improve the antioxidant status of cultured L. vannamei. In this study, increasing the availability of minerals as cofactors for antioxidant enzymes can be one of the possible reasons for promoting the antioxidant status in supplemented groups.
Increasing the survival rate against fatal infections is one of the main goals of adding biostimulants to aquafeeds. In this study, the diets fortified with TA at all concentrations promoted a significant decrease in the mortality rate of the shrimp compared to animals given the TA0 treatment. Moreover, the minimum MR was obtained in the TA 7.5 group, which was three times lower than the control group. This can be related to the better regulation of the intestine microflora, increasing the immune and antioxidant capacity of shrimp in this experimental treatment. Other researchers have reported similar, enhanced resistance against V. parahaemolyticus in L. vannamei fed diets with formic acid, and citric acid plus sorbic acid.
Perspectives
Results of this study showed that dietary TA improved the growth markers and defense systems of L. vannamei. The notable improvements in growth indices, feed utilization, digestive enzyme activities, hemolymph immune parameters, antioxidant status, and disease resistance against V. parahaemolyticus were mostly recorded in the shrimp fed with the TA7.5 diet.
These results showed that TA could be considered a novel immunopotentiator and growth stimulator in diets for cultured L. vannamei. However, more research is recommended to explore the properties of tartaric acid alone or in combination with other biostimulants – such as probiotics – on gut morphology, the expression of genes related to growth, immunity, and antioxidant capacity, as well as its benefits against environmental stresses in farmed species of fish and shellfish.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Morteza Yousefi, Ph.D.
Corresponding author
Department of Veterinary Medicine, RUDN University, Moscow, Russia[109,111,99,46,108,105,97,109,103,64,49,56,105,102,101,115,117,111,121,109]
Tagged With
Related Posts

Intelligence
Playing favorites: How selective breeding can grow the seaweed sector
Seaweed farming experts are urging producers to employ selective breeding to capitalize on desired traits amid a changing climate.

Health & Welfare
Biofloc potential to alter virulence of V. parahaemolyticus strain on L. vannamei postlarvae
A biofloc environment has the potential to alter the virulence of the AHPND-causing V. parahaemolyticus strain on Pacific white shrimp postlarvae.

Health & Welfare
Dietary potassium diformate improves pangasius growth performance, yield
In a laboratory trial, juvenile pangasius given a diet with 0.2 percent potassium diformate (KDF) had better weight gain, feed conversion and survival than fish that did not receive KDF.

Health & Welfare
Estimating heritability of WSSV resistance in Pacific white shrimp
A study in Vietnam showed that there was significant variation in WSSV resistance among Pacific white shrimp families evaluated.


![Ad for [Aquademia]](https://www.globalseafood.org/wp-content/uploads/2025/07/aquademia_web2025_1050x125.gif)
