High-quality feeds, feed monitoring, water quality management should be high priorities

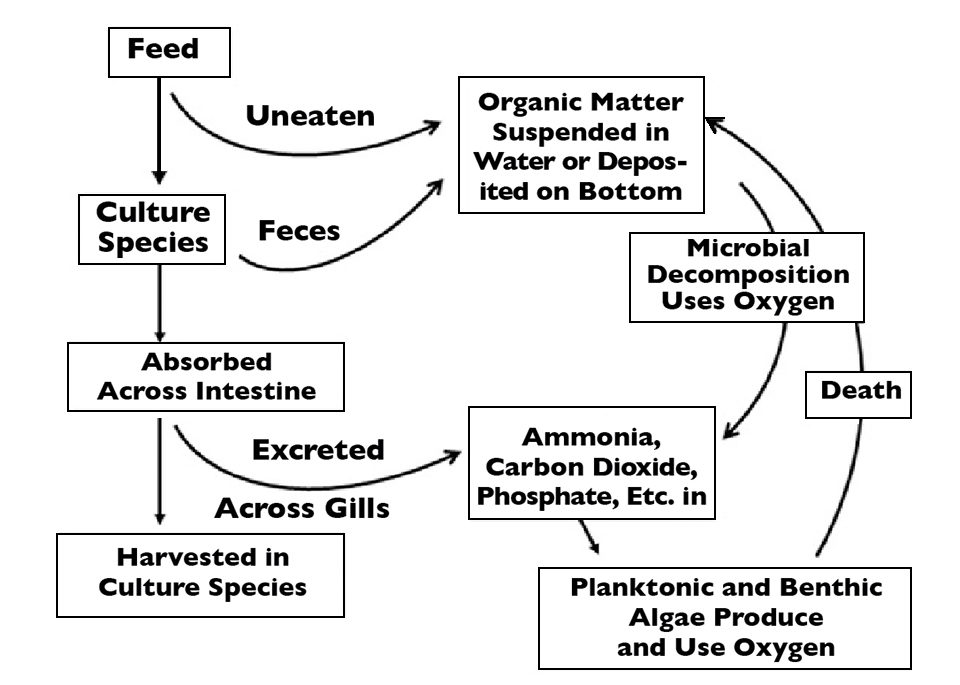
The fate of feed applied to aquaculture systems is illustrated in Fig. 1. A portion of the feed is either not eaten or expelled as feces. This part is decomposed by bacteria that consume oxygen and excrete ammonia nitrogen in the process.
Feed nutrients absorbed by the culture species are used in respiration, excreted as ammonia nitrogen and other metabolites, or converted to biomass and removed from the culture system at harvest. Generally, 8 to 12 percent of the organic carbon and 20 to 30 percent of the nitrogen contained in feed are removed in biomass at harvest.
Dissolved oxygen removed from the water by respiration of culture animals, microbial decomposition of uneaten feed and feces, and oxidation of ammonia nitrogen by nitrifying bacteria represent the oxygen demand of the feed. Much of the oxygen demand is expressed in the pond during the culture period.
However, some of the oxygen demand is deferred until later because some of the organic carbon from feeding waste is stored in sediment organic matter. A portion of the demand is also externalized by discharge into natural water bodies through overflow or water exchange, and when ponds are drained for harvest.
Oxygen demand of feed
Nutrients from feed stimulate phytoplankton photosynthesis that produces dissolved oxygen and organic matter in ponds. The amount of oxygen produced in photosynthesis is sufficient to oxidize the organic carbon produced in the process, and the oxygen demand of feed should not include the organic matter produced by photosynthesis.
The oxidation of organic carbon requires 2.67 units of molecular oxygen per unit organic carbon regardless of whether oxidation is by bacteria or the culture species. The oxidation of ammonia nitrogen to nitrate nitrogen by nitrifying bacteria requires 4.57 units of molecular oxygen per unit of nitrogen.
Shrimp ponds
To illustrate the calculation of the oxygen demand of feed (Fig. 2), assume that the feed-conversion efficiency (FCE) in a shrimp pond is 0.556, feed-conversion ratio (FCR) is 1.8, feed contains 6 percent nitrogen and 45 percent carbon, and shrimp contain 2.75 percent nitrogen and 11 percent carbon. One kilogram of the feed will produce 0.556 kg shrimp. This amount of feed contains 0.06 kg nitrogen and 0.45 kg carbon, and the shrimp produced with it contain 0.015 kg nitrogen and 0.0612 kg carbon.
Subtracting the carbon and nitrogen in 0.556 kg shrimp from that in 1 kg feed gives a waste load of 0.045 kg nitrogen and 0.389 kg carbon. The oxygen demand of the feed is (0.0045 kg nitrogen × 4.57) + (0.389 kg carbon × 2.67) = 1.245 kg oxygen per kg feed. These computations can be arranged into the equation in Fig. 2.

The oxygen demand of feed varies with FCE as illustrated in Table 1 using feed and shrimp composition data from the example above. Notice that FCE does not make a large difference in the oxygen demand per kilogram of feed. Moreover, there is not a large variation in nitrogen and carbon percentages in feed and shrimp.
Therefore, a value of 1.25 kg oxygen per kg feed would seem to be reasonable for general use. Feed-conversion efficiencies, feed composition and body composition do not vary tremendously for most warmwater culture species. Thus, an oxygen demand of 1.25 kg oxygen per kg feed also can be used for most types of warm-water aquaculture.
The oxygen demand caused by feed added to the culture system, however, is strongly influenced by FCE as also illustrated in Table 1. Improving the FCE from 0.417 to 0.625, which lowers the FCR from 2.4 to 1.6, reduces the feed oxygen demand by 1.18 kg oxygen per kg shrimp – a reduction of 37.8 percent.
Boyd, Feed efficiency and oxygen demand, Table 1
| Feed-Conversion Efficiency | Feed-Conversion Ratio | Oxygen Demand of Feed (kg oxygen/ kg feed) | Oxygen Demand of Feed to Produce 1 kg Shrimp (kg oxygen/kg shrimp) |
|---|---|---|---|
| 0.625 | 1.6 | 1.214 | 1.94 |
| 0.566 | 1.8 | 1.245 | 2.24 |
| 0.500 | 2.0 | 1.266 | 2.53 |
| 0.455 | 2.2 | 1.285 | 2.83 |
| 0.417 | 2.4 | 1.301 | 3.12 |
Such a large reduction in oxygen demand would improve water quality in ponds. It also would lessen the oxygen demand of effluents discharged from ponds. Equally important, it takes 0.8 kg less feed to produce 1 kg shrimp at an FCR of 1.6 than at 2.4 – a considerable economic advantage. Thus, implementation of measures such as using high-quality feeds, adopting feeding practices to assure that most applied feed is eaten, and practicing good water quality management that can reduce FCR should be given high priority by producers.
Much of the feed oxygen demand is exerted in ponds. In flowing water systems, uneaten feed and feces often are partially removed by sedimentation before final effluent discharge. Therefore, the feed oxygen demand is not a substitute for the biological oxygen demand of effluent from ponds and flow-through systems.
In cage and net pen culture, all carbon and nitrogen not removed in fish biomass enters the ecosystems containing the cages or net pens. The oxygen demand exerted on the ecosystem can be estimated as follows:

Tilapia, salmon cages
A tilapia cage culture operation in a lake that produces 10,000 metric tons (MT) annually at an FCR of 1.7 will use 17,000 MT feed per year. Assuming the feed oxygen demand is about 1.25 kg oxygen per kg, the oxygen demand imposed upon the ecosystem would be 21,250 MT per year.
Much lower FCRs are typically achieved in salmon culture than in tilapia culture. Approximate calculations suggest that the oxygen demand of salmon feed in a system with an FCR of 1.1 also would be about 1.25 kg oxygen per kg feed. Because of the lower FCR, salmon cage culture would result in a smaller oxygen demand upon the surrounding ecosystem than caused by production of an equal quantity of tilapia in cages.
(Editor’s Note: This article was originally published in the November/December 2009 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-

Claude E. Boyd, Ph.D.
Department of Fisheries and Allied Aquacultures
Auburn University
Alabama 36849 USA[117,100,101,46,110,114,117,98,117,97,64,49,101,99,100,121,111,98]
Tagged With
Related Posts

Responsibility
A look at unit processes in RAS systems
The ability to maintain adequate oxygen levels can be a limiting factor in carrying capacities for RAS. The amount of oxygen required is largely dictated by the feed rate and length of time waste solids remain within the systems.

Responsibility
A look at various intensive shrimp farming systems in Asia
The impact of diseases led some Asian shrimp farming countries to develop biofloc and recirculation aquaculture system (RAS) production technologies. Treating incoming water for culture operations and wastewater treatment are biosecurity measures for disease prevention and control.

Intelligence
Adding value to tilapia to tap into U.S. market
New markets for tilapia and expansion of existing ones can be created by planning and implementing properly designed geographic strategies to meet discriminating consumer preferences. Low labor costs in most producing countries promotes value-adding by the production of fresh fillets.

Responsibility
Appraising pond liners for shrimp culture
The use of plastic-lined ponds by shrimp farmers can significantly improve production efficiency, support more production cycles per year, and higher mechanical aeration rates and stocking densities. The capital cost of lining ponds can be very significant, so a thorough feasibility analysis is recommended when considering this production tool.

