Producers must balance the benefits with risks of overuse

Fertilizers frequently are used in pond aquaculture to stimulate phytoplankton productivity and enhance the availability of natural food organisms. Turbidity created by the phytoplankton also shades pond bottoms to discourage the growth of aquatic weeds.
Fertilizers are a valuable and expensive resource that should be used wisely and conservatively. Excess use of fertilizer is wasteful, encourages an overabundance of phytoplankton, and increases the concentrations of nutrients in aquaculture effluents.
Nutritive elements
Plants require nutritive elements that include oxygen, hydrogen, carbon, phosphorus, nitrogen, sulfur, calcium, magnesium, potassium, sodium, iron, manganese, zinc, and copper. Some species also may need boron, cobalt, molybdenum, and other trace elements.
In most soils, it is only necessary to apply nitrogen, phosphorus, and potassium to promote plant growth. These three nutrients are called primary nutrients in fertilizers. In aquaculture ponds, potassium fertilization usually is not necessary, so nitrogen and phosphorus are the most important nutrients.
Common fertilizers
The two most common fertilizers used worldwide in aquaculture are triple superphosphate (TSP) and urea. TSP is made by treating rock phosphate with phosphoric acid. Urea is made by reacting ammonia and carbon dioxide. Other common fertilizers used in agriculture and aquaculture are listed in Table 1.
Boyd, Approximate grades of common commercial fertilizers, Table 1
| Fertilizer | Primary Nutrients (%) N | Primary Nutrients (%) P2O5 | Primary Nutrients (%) K2O |
|---|---|---|---|
| Urea | 45 | 0 | 0 |
| Calcium nitrate | 15 | 0 | 0 |
| Sodium nitrate | 16 | 0 | 0 |
| Ammonium nitrate | 33-35 | 0 | 0 |
| Phosphoric acid | 20-21 | 0 | 0 |
| Phosphoric acid | 0 | 52-60 | 0 |
| Ammonium polyphosphate | 10-13 | 30-38 | 0 |
| Superphosphate | 0 | 18-20 | 0 |
| Triple superphosphate | 0 | 44-54 | 0 |
| Monoammonium phosphate | 11 | 48 | 0 |
| Diammonium phosphate | 18 | 48 | 0 |
| Potassium phosphate | 0 | 52 | 34 |
| Calcium metaphosphate | 0 | 62-64 | 0 |
| Potassium nitrate | 13 | 0 | 44 |
| Potassium sulfate | 0 | 0 | 50 |
| Muriate of potash | 0 | 0 | 60 |
Muriate of potash and sodium nitrate are extracted from ores. The other materials are made from the raw materials of rock phosphate and ammonia. Rock phosphate is a phosphate-bearing mineral, but ammonia is made industrially by reacting atmospheric nitrogen with hydrogen. Most fertilizers are packed in bags and sold as dry granules or prills, but ammonium polyphosphate and phosphoric acid are liquids.
In fertilizers, nitrogen is present as urea, ammonium, or nitrate. Phosphorus occurs as orthophosphate or polyphosphate, while potassium appears in its ionic form. Fertilizers dissolve in water to release their nutrients. Urea begins to hydrolyze at once and is completely transformed to ammonia and carbon dioxide within hours or days. Polyphosphate also quickly hydrolyzes to orthophosphate.
Fertilizer grades
The grade of a fertilizer usually is reported as percentages of nitrogen (N), phosphorus oxide (P2O5), and potassium oxide (K2O). Thus, triple superphosphate usually is a 0-46-0 fertilizer, diammonium phosphate is a 18-48-0 fertilizer, and urea is a 45-0-0 fertilizer. Table 1 shows the approximate grades of common commercial fertilizers. Notice that some primary fertilizer sources contain one primary nutrient, such as urea or triple superphosphate, while others contain two primary nutrients like diammonium phosphate and potassium phosphate.
Fertilizer nutrient content is sometimes reported in elemental form, i.e., nitrogen, phosphorus (P), and potassium (K). The factor for converting P2O5 to P is 0.437, while the factor for converting K2O to K is 0.83. For example, triple superphosphate usually is about 46 percent P2O5. The P content is 46 percent P2O5 x 0.437 = 20.1 percent P. The P percentage can be converted to P2O5 by dividing the P content by 0.437.
Mixed fertilizers
A mixed fertilizer is made by blending two or more primary fertilizer sources to provide two or three primary nutrients. For example, a popular fish pond fertilizer used inthe United States has a grade of 20-20-5. It can be made byblending several combinations of fertilizer sources, one ofwhich is urea, triple superphosphate, and muriate of potash(MP). A 100-kg quantity would contain 20 kg each of Nand P2O5, and 5 kg K2O, and could be made as follows:

The nutrient sources do not add up to 100 kg, so a fillersuch as agricultural limestone must be added:

Mixed fertilizers representing a wide range of grades can be purchased in some nations.
Secondary nutrients
Sometimes fertilizers are supplemented with the secondary nutrients calcium, magnesium, and sulfur. The usual sources of these elements are calcium and magnesium sulfates. Supplements of trace elements, iron, manganese, zinc, copper, boron, and others may also be added to fertilizers.
Metallic trace elements should be chelated to enhance solubility and availability to phytoplankton. Trace elements combined with citric acid, ethylenediaminetetraacetic-acid (EDTA), or other chelating agents can be added, but inclusion of chelated trace metals increases fertilizer costs appreciably.
Liquid fertilizers
Fertilizer granules or prills are water-soluble but settle to the pond bottom before completely dissolving. Much of their phosphorus can be adsorbed by the bottom soil instead of dissolving in the water. This problem can be lessened by using liquid fertilizers.
Liquid fertilizers are denser than water and should be diluted 1:10 with pond water and splashed over pond surfaces or released into the propeller wash of an outboard motor while a boat is driven over the pond surface. If liquid fertilizers are not available or considered too expensive, granular or prilled fertilizers can be placed in 10-20 times their volume of pond water, predissolved by vigorous stirring, and splashed over the water surface.
Application rates
Application rates for N and P2O5 are usually 2-10 kilograms per hectare per application. Many farmers apply excessive nitrogen, however. I recommend applications of 2 kg N per hectare and 8 kg P2O5 per hectare for freshwater ponds, and 8 kilogram per hectare each for N and P2O5 in ponds filled with brackish water or seawater.
Fertilizer may need to be applied at intervals of two to four weeks to maintain phytoplankton blooms. However, more frequent applications may be necessary to initiate blooms. In ponds with feed applications, nutrients enter the water from feed wastes, and applications of fertilizers often are unnecessary or only necessary until feeding rates reach 15-20 kilograms per hectare per day.
Once blooms have been initiated, fertilizer can be applied according to need as judged by Secchi disk visibility. A Secchi disk visibility of 25-40 cm usually is considered adequate. As an example, the following scale can be used where normal application rates are 2 kg N per hectare and 8 kg P2O5 per hectare.
Boyd, Table 2
| Secchi Disk Visibility (cm) | Application Rate (kg/ha) N | Application Rate (kg/ha) P2O5 |
|---|---|---|
| 25 or less | 0 | 0 |
| 26-30 | 0.5 | 2 |
| 31-35 | 1.0 | 4 |
| 36-40 | 1.5 | 6 |
| More than 40 | 2 | 8 |
Most fish and shrimp farmers do not use mixed fertilizers, but purchase primary fertilizer sources. The application rates of urea (45 percent) and triple superphosphate (46 percent P2O5) necessary to provide 2 kilograms per hectare N and 8 kilograms per hectare P2O5 can be estimated as follows:
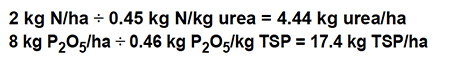
Of course, if a mixed fertilizer is used as an alternative to urea and triple superphosphate in the example above, it should have a ratio of 1 N:4 P2O5. This could be achieved with a 5-20-0 fertilizer or 8-32-0 fertilizer.
Diammonium phosphate and monoammonium phosphate are good fertilizers for freshwater, because they have N:P2O5 ratios of about 1:3 and 1:4, respectively. For brackish water or seawater, a fertilizer with a 1:1 N:P2O5 ratio is best, and a mixed fertilizer with a 20-20-0 grade is a good choice.
In brackish ponds used for shrimp culture, many farmers want to encourage diatom growth. This can be done by applying nitrogen fertilizers weekly at 2-3 kg N per hectare. Nitrate is especially efficient in promoting diatoms, and sodium nitrate can be used as a diatom-promoting fertilizer.
Fertilizers containing ammonium or urea are potentially acidic because nitrifying bacteria in water can oxidize ammonia to nitrate and yield hydrogen ions as shown in the following equation for ammonium sulfate:

The hydrogen ions react in water to decrease total alkalinityand pH. The adverse influence of nitrification on alkalinityand pH can be counteracted by routine applicationsof agricultural limestone to ponds where alkalinity is naturallylow. In brackish water, seawater, and freshwater withalkalinity above 50 milligram per liter, acidity from nitrogen fertilizersusually is not a problem.
Storage
Fertilizers should be stored in a dry place to prevent caking. Since nitrate fertilizers are strong oxidants and explosive, they should not be stored near oil products or where sparks occur. Environment spills should be avoided to prevent toxicity to aquatic life and nutrient pollution.
(Editor’s Note: This article was originally published in the June 2003 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-

Claude E. Boyd, Ph.D.
Professor, Department of Fisheries and Allied Aquacultures
Auburn University
Auburn, Alabama 36849 USA[117,100,101,46,110,114,117,98,117,97,46,103,97,115,101,99,97,64,100,121,111,98,101,99]
Tagged With
Related Posts

Responsibility
Accuracy of custom water analyses varies
The reliability of trace element analyses reported by custom laboratories cannot be checked by simple techniques, and results may not always be accurate. One should check the reliability of major ion analyses by determining the charge balance and comparing the measured total ion concentration with the total ion concentration estimated from conductivity.

Responsibility
Defining ranges for water quality variables presents complex, challenging process
The creation and application of reference tables for acceptable concentration ranges of physical and chemical water quality variables for culture organisms would be challenging due to the differing tolerances found among the many farmed species.

Health & Welfare
Decomposition and accumulation of organic matter in ponds
Aquaculture ponds accumulate organic matter from organic fertilizer, remains of microorganisms produced within the pond, feces of the culture animals and uneaten feed. Claude E. Boyd, Ph.D., details the leading organic matter management practices, and says that the accumulation of organic matter is often not as great as believed.

Innovation & Investment
After half a century, Auburn’s Claude Boyd to ‘retire’
Friends, colleagues and former students of longtime Auburn University Professor Claude E. Boyd, Ph.D., don’t expect the Mississippi native to slow down, despite the quiet announcement of his retirement earlier this month. The 50-year veteran researcher’s career has been truly extraordinary.


![Ad for [BSP]](https://www.globalseafood.org/wp-content/uploads/2025/07/BSP_B2B_2025_1050x125.jpg)